Complete the following reactions.
(a)
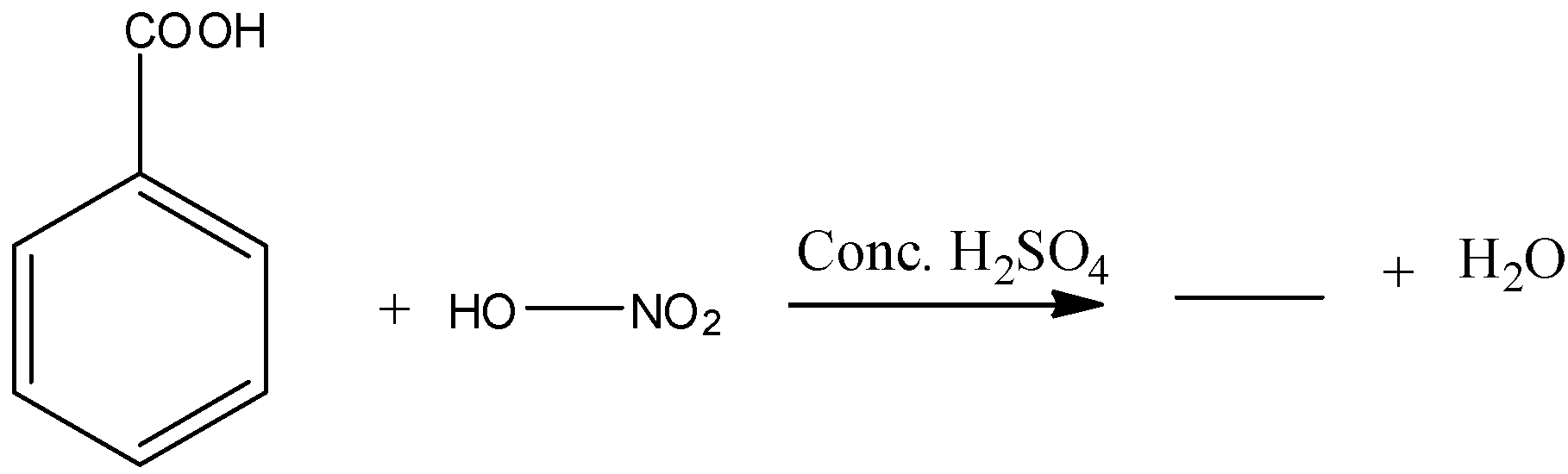
(b)
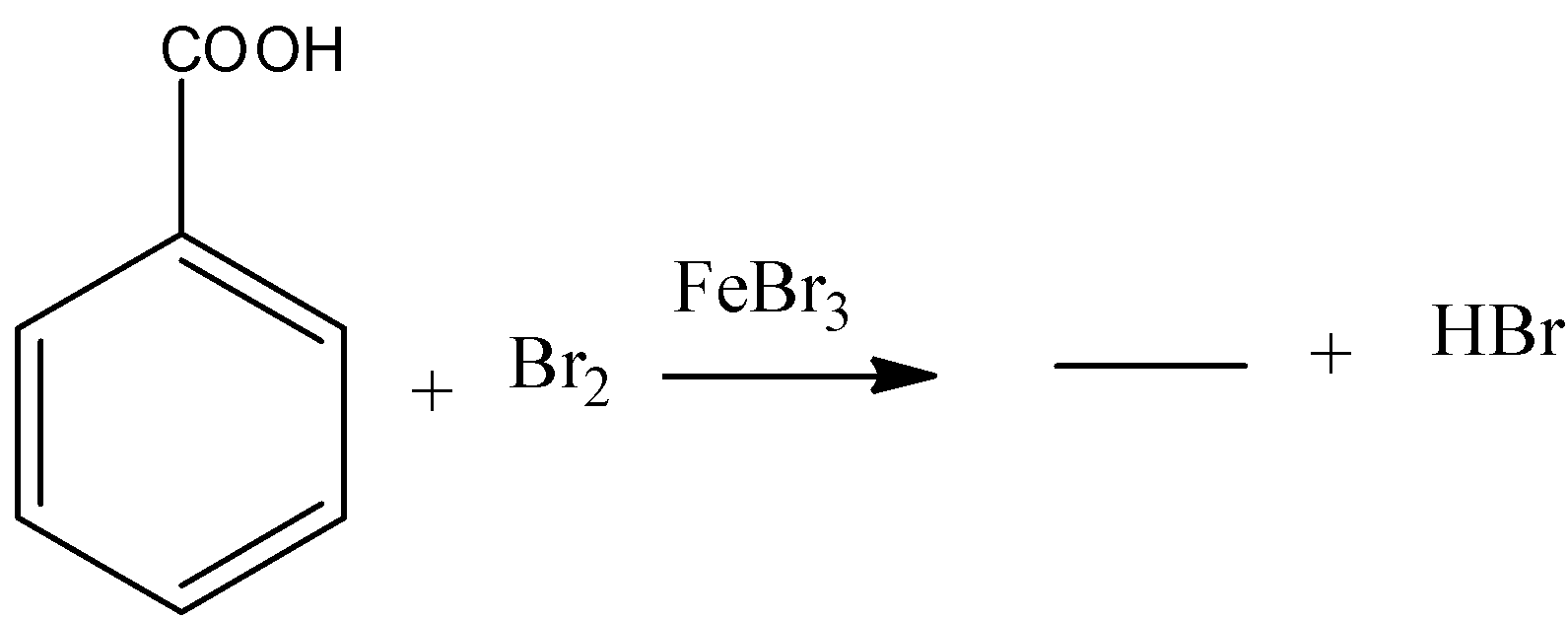
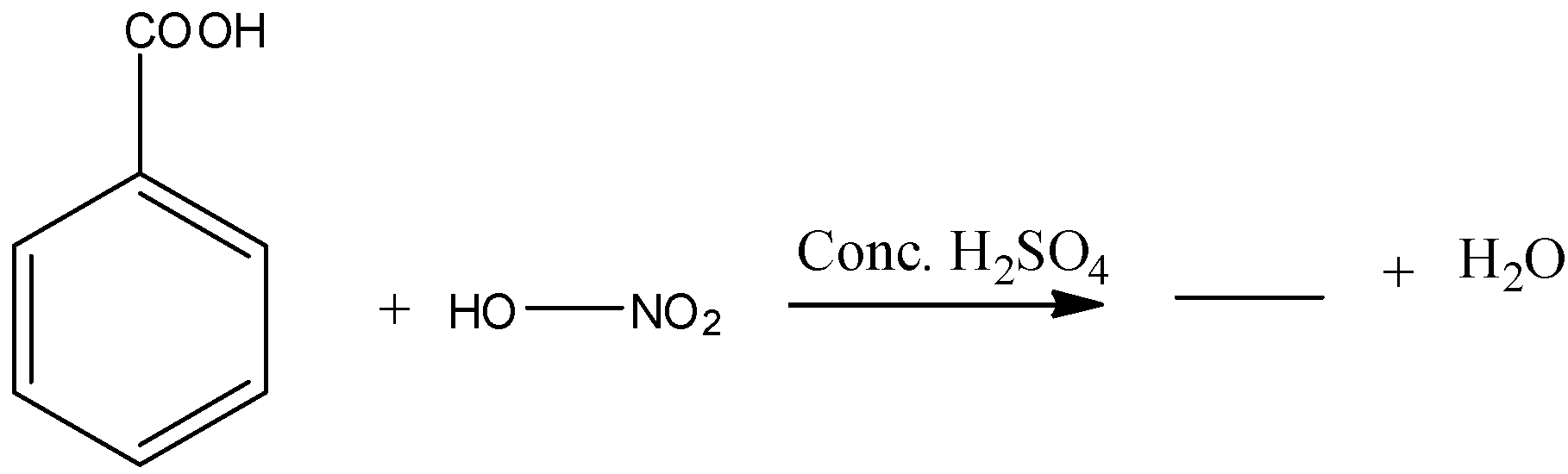

Answer
598.2k+ views
Hint: The presence of the electron withdrawing group directs the reactants to meta position that is why electron withdrawing groups are called meta directing groups. Carboxylic acid comes under electron withdrawing groups and acts as a meta directing group.
Complete step by step answer:
- In the question it is given to complete the chemical reactions.
a) The given chemical reaction is as follows.
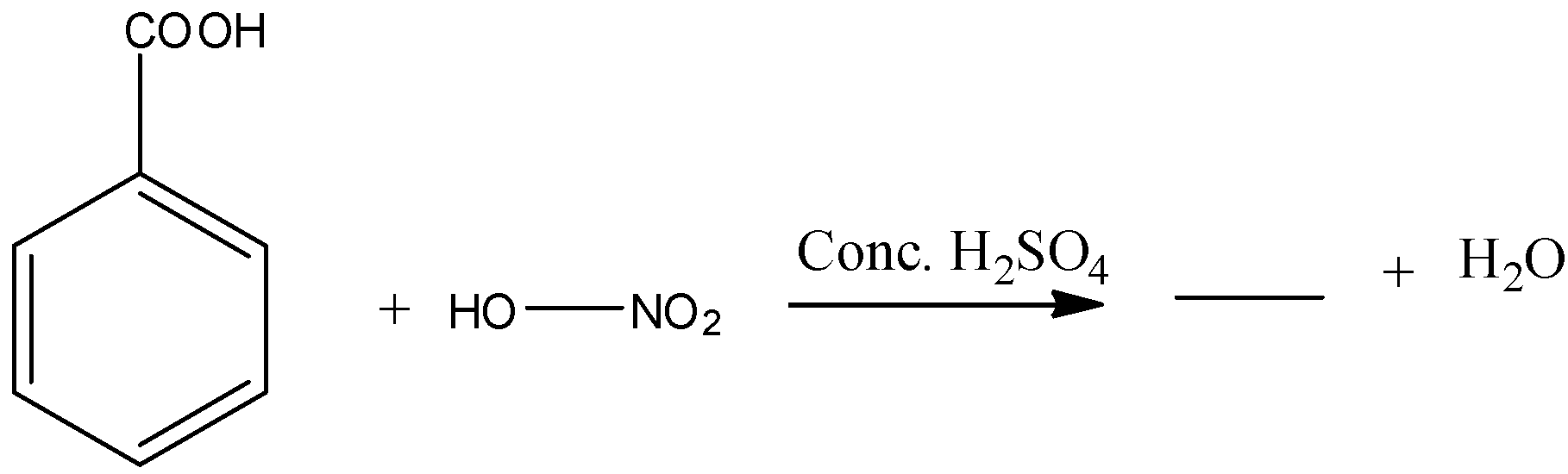
- In the above chemical reaction benzoic acid reacts with nitric acid in presence of sulphuric acid.
- The chemical reaction of benzoic acid reacts with nitric acid in presence of sulphuric acid is as follows.
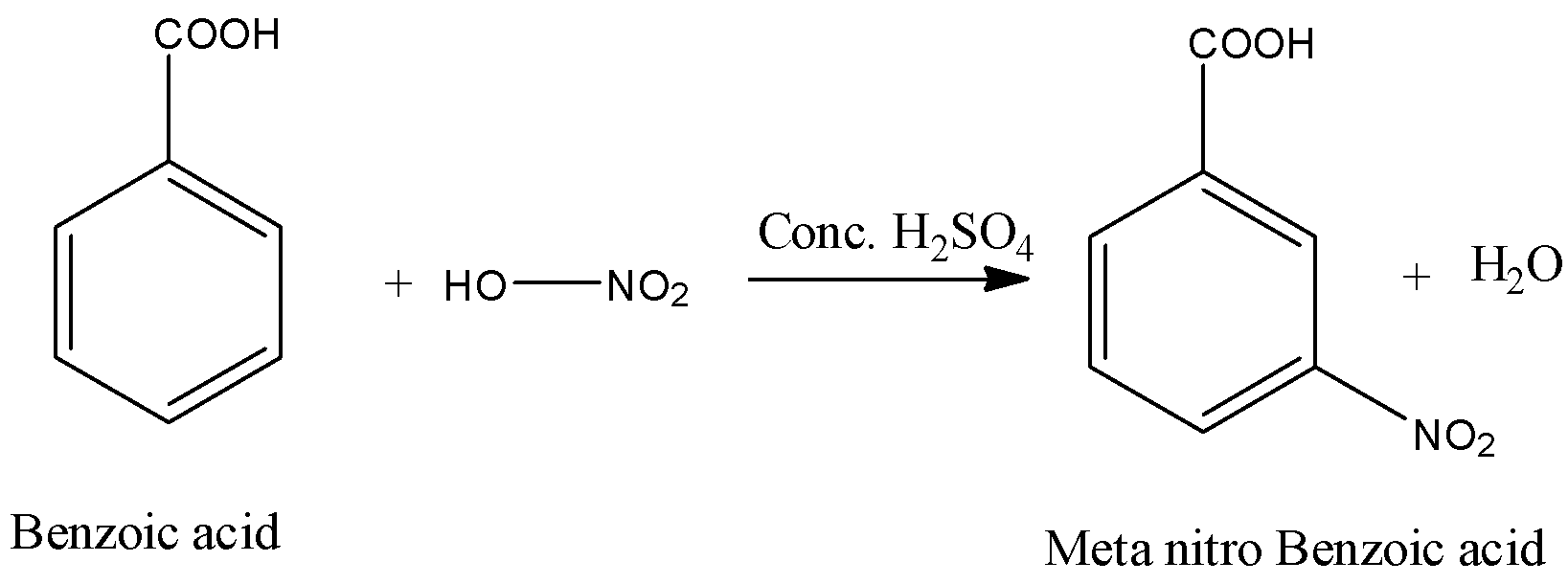
- In the above chemical reaction nitric acid reacts with benzoic acid and forms meta nitro benzoic acid and water as the products.
b) The given chemical reaction is as follows.
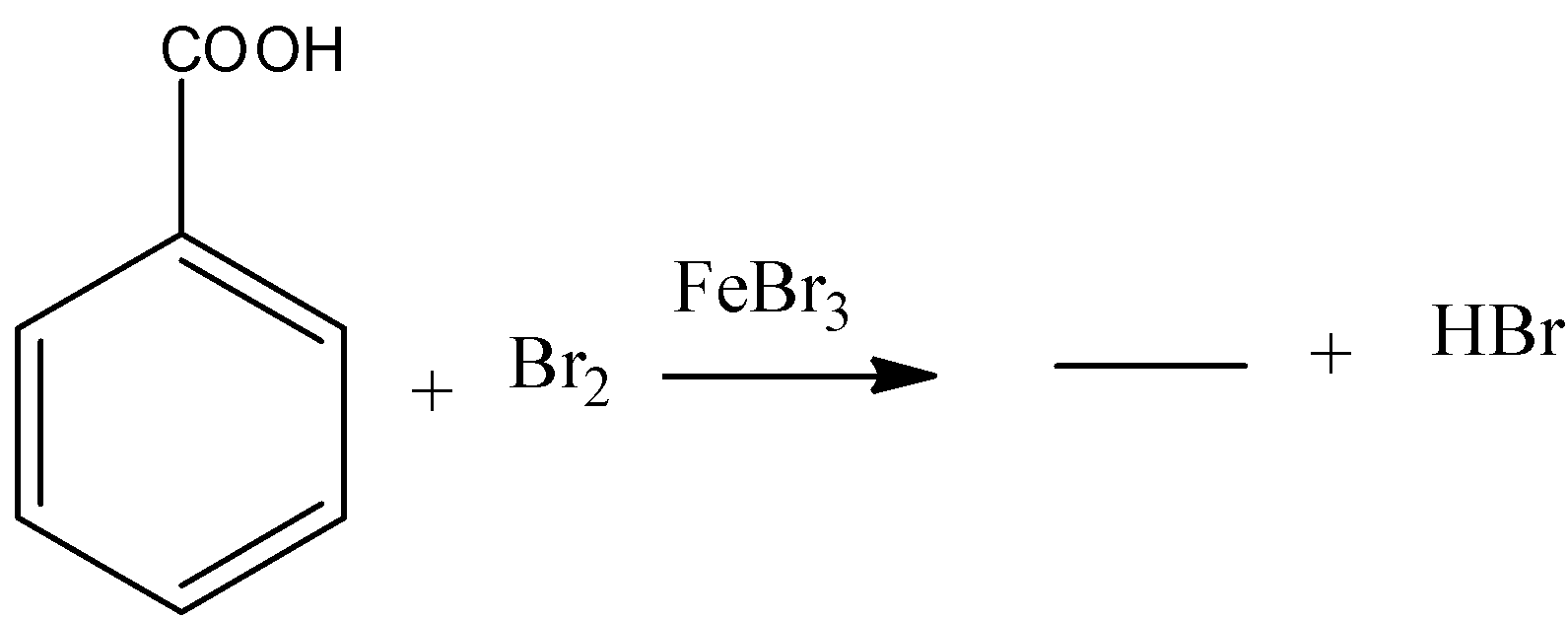
- In the above chemical reaction benzoic acid reacts with bromine in presence of ferric bromide reagent.
- The chemical reaction of benzoic acid reacts with bromine in presence of ferric bromide is as follows
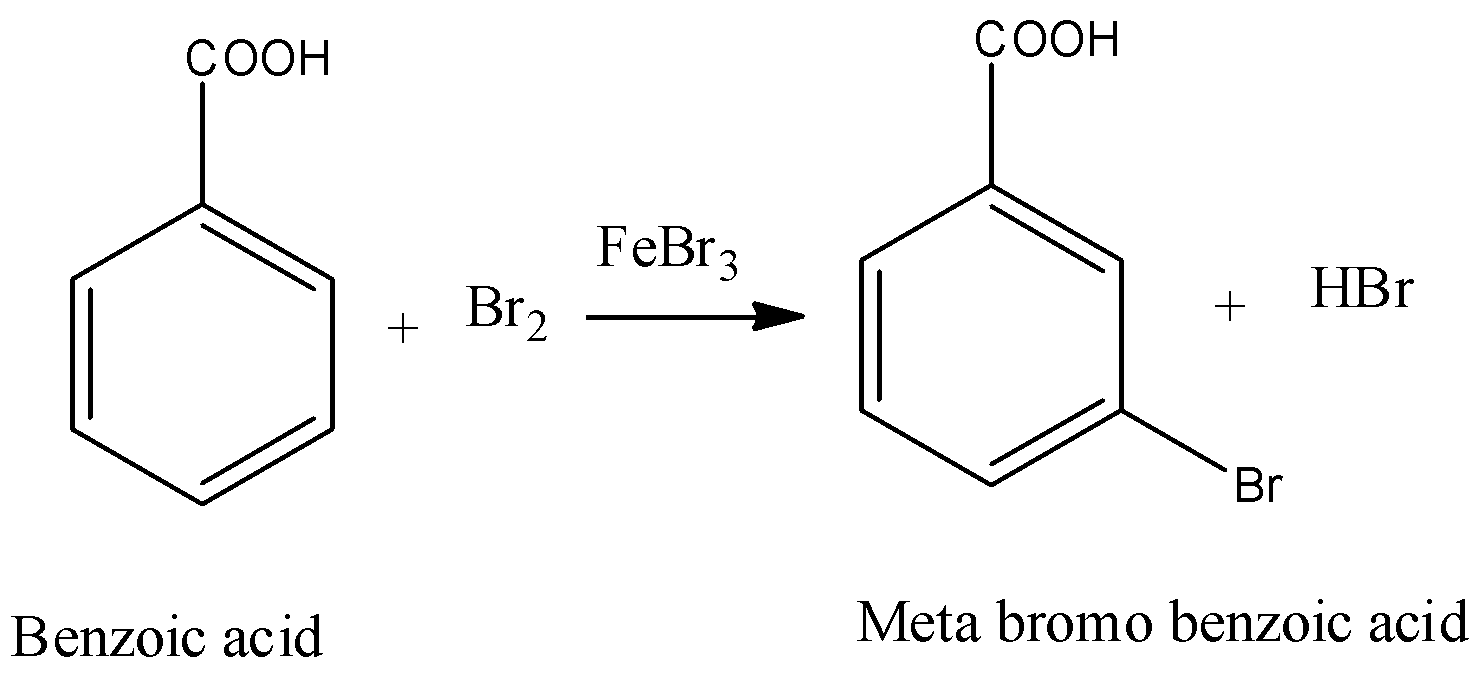
- In the above chemical reaction bromine reacts with benzoic acid and forms meta bromo benzoic acid and hydrogen bromide as the products.
Note: If we are going to add a nitro functional group in the benzene then it is called nitration. If we are going to add bromine in the benzene ring then it is called bromination or halogenation. In presence of the electron withdrawing group in benzene nitration and bromination occurs at meta position in the benzene ring.
Complete step by step answer:
- In the question it is given to complete the chemical reactions.
a) The given chemical reaction is as follows.
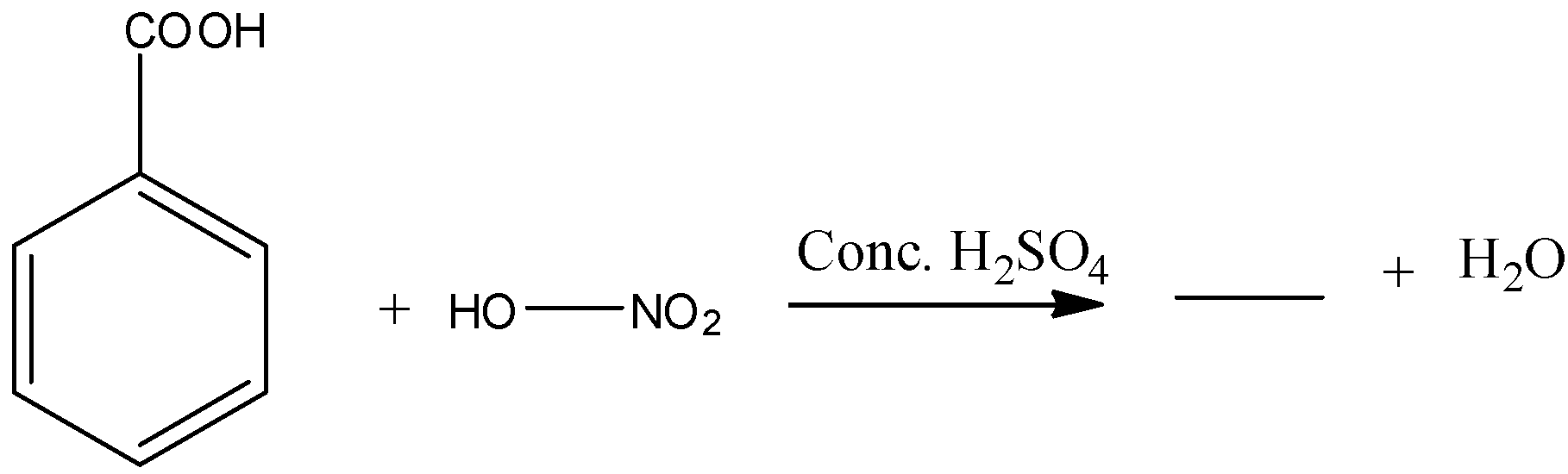
- In the above chemical reaction benzoic acid reacts with nitric acid in presence of sulphuric acid.
- The chemical reaction of benzoic acid reacts with nitric acid in presence of sulphuric acid is as follows.

- In the above chemical reaction nitric acid reacts with benzoic acid and forms meta nitro benzoic acid and water as the products.
b) The given chemical reaction is as follows.

- In the above chemical reaction benzoic acid reacts with bromine in presence of ferric bromide reagent.
- The chemical reaction of benzoic acid reacts with bromine in presence of ferric bromide is as follows

- In the above chemical reaction bromine reacts with benzoic acid and forms meta bromo benzoic acid and hydrogen bromide as the products.
Note: If we are going to add a nitro functional group in the benzene then it is called nitration. If we are going to add bromine in the benzene ring then it is called bromination or halogenation. In presence of the electron withdrawing group in benzene nitration and bromination occurs at meta position in the benzene ring.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




