\[Cl - P - Cl\] bond angles in $PC{l_5}$ molecule is:
A. ${120^ \circ }$ and ${90^ \circ }$
B. ${60^ \circ }$ and ${90^ \circ }$
C. ${60^ \circ }$ and ${120^ \circ }$
D. ${120^ \circ }$ and ${30^ \circ }$
Answer
597k+ views
Hint: Bond angles contribute the shape of a molecule. Bond angles are the angles between adjacent lines that represent bonds. The bond angle can help differentiate between linear, trigonal planar, tetrahedral, trigonal bipyramidal and octahedral. It depends on the number of lone electron pairs. It is determined by the lone pair electrons repelling the bonding pairs until the ligands. Bond angles are used to little trigonometry to calculate how far apart the ligands themselves are.
Complete step by step:
According to VSEPR theory, \[P - Cl\] is bonding environments and forms two bonds in this molecule. Here we see the structure of $PC{l_5}$ which is given below:
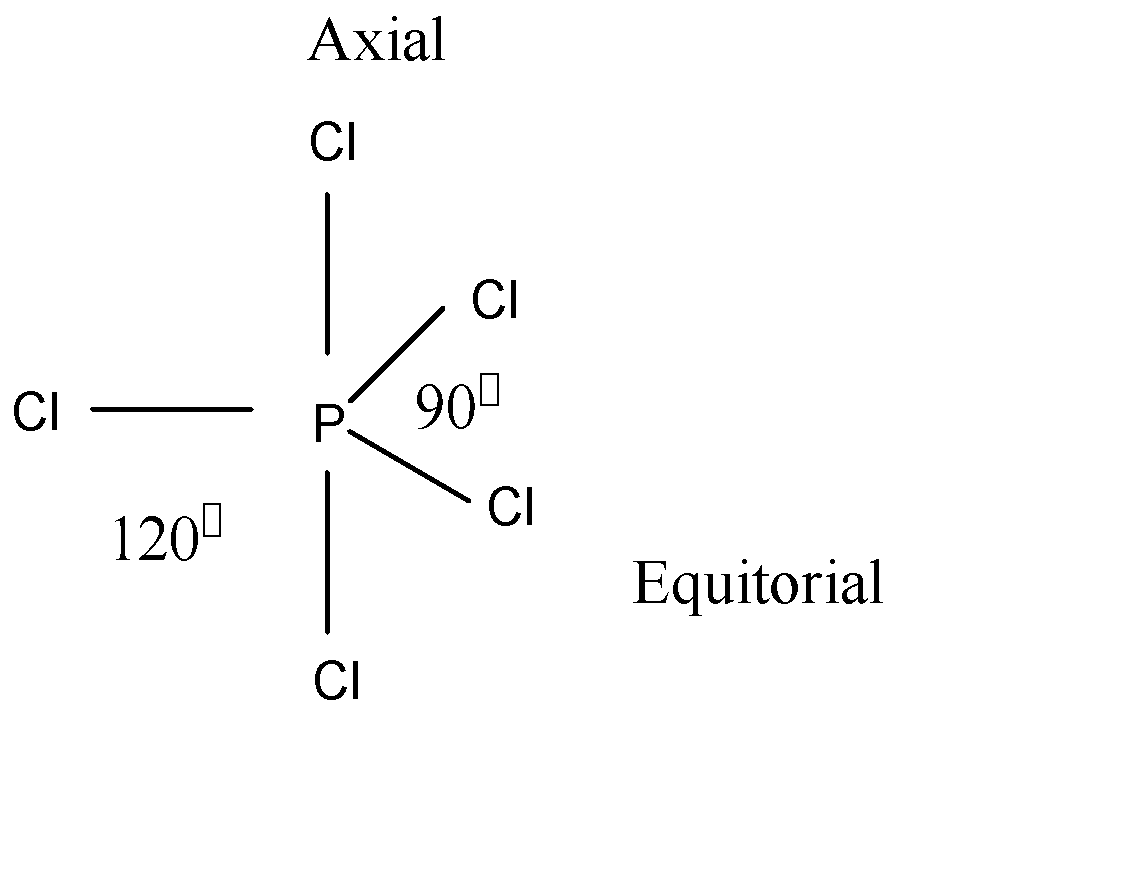
Here the central atom is P and it contributes 5 electrons. As we see the structure, there is 5 Cl to bond with P. So 5 Cl is contributing a total of 5 electrons. So, P and Cl contribute a total 10 electrons. The geometry of $PC{l_5}$ is trigonal bipyramidal.
Here each two \[P - Cl\] bond makes two ${90^ \circ }$ and two ${120^ \circ }$ bond angles with the other bonds in the molecule.
Hence, option (A) is the correct answer.
Note: Here we must remember that the axial to the three equatorial bonds, so one axial bond is at right angle to 3 bonds. Therefore, 2 axial bonds will be at the right angle to 6 bonds. So, the number of right angles is 6. Each equatorial $PC{l_5}$ makes two right angles. The mixing of ones, three P and one d atomic orbitals to form five $s{p^3}d$ hybrid orbitals of equal energy is called $s{p^3}d$ hybridization.
Complete step by step:
According to VSEPR theory, \[P - Cl\] is bonding environments and forms two bonds in this molecule. Here we see the structure of $PC{l_5}$ which is given below:

Here the central atom is P and it contributes 5 electrons. As we see the structure, there is 5 Cl to bond with P. So 5 Cl is contributing a total of 5 electrons. So, P and Cl contribute a total 10 electrons. The geometry of $PC{l_5}$ is trigonal bipyramidal.
Here each two \[P - Cl\] bond makes two ${90^ \circ }$ and two ${120^ \circ }$ bond angles with the other bonds in the molecule.
Hence, option (A) is the correct answer.
Note: Here we must remember that the axial to the three equatorial bonds, so one axial bond is at right angle to 3 bonds. Therefore, 2 axial bonds will be at the right angle to 6 bonds. So, the number of right angles is 6. Each equatorial $PC{l_5}$ makes two right angles. The mixing of ones, three P and one d atomic orbitals to form five $s{p^3}d$ hybrid orbitals of equal energy is called $s{p^3}d$ hybridization.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Which gas is abundant in air class 11 chemistry CBSE

Explain zero factorial class 11 maths CBSE

Difference between unicellular and multicellular o class 11 biology CBSE

What organs are located on the left side of your body class 11 biology CBSE

Why is decomposition of vegetable matter into compost class 11 chemistry CBSE




