Choose the correct option for - which is not the structure of $ {C_4}{H_9}Br $
(A) $ 1 - $ Bromobutane
(B) $ 2 - $ Bromobutane
(C) $ 1 - $ Bromo - $ 2 - $ methylpropane
(D) Isobutane
Answer
554.1k+ views
Hint :Bromobutane is a colourless liquid, but its impure sample can be seen as slightly yellow in colour. Bromobutane is an organobromine compound with a molecular formula of $ {C_4}{H_9}Br $ and the boiling point of around $ 100 - {205^o}C $ .
Complete Step By Step Answer:
To solve this question we should have some insight regarding the conformation of structures.
Looking at our options - $ 1 - $ Bromobutane and $ 2 - $ Bromobutane, let’s have a look at their structure for the better understanding –
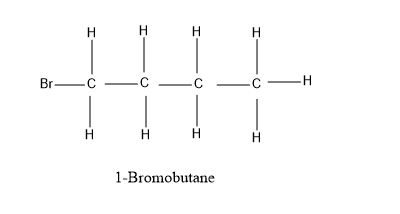
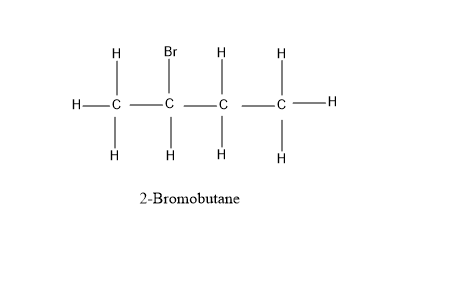
As, it is evident from the diagram above, that $ 1 - $ Bromobutane and $ 2 - $ Bromobutane fits completely into the formula $ {C_4}{H_9}Br $ . All the valencies of all the atoms are completely satisfied and have a stable structure under this formula - $ {C_4}{H_9}Br $ .
Similarly. $ 1 - $ Bromo - $ 2 - $ methyl propane – also has a stable structure satisfying all the valences of each atom.
But, coming to our option D , it is isobutane . It has a molecular formula of $ {C_4}{H_{10}} $ . Isobutane does not consist of bromine atoms. So it does not fit in our given formula $ {C_4}{H_9}Br $ .
So, the correct option to our question is D i.e. isobutane.
Note :
For $ {C_4}{H_9}Br $ we have four possible structural isomers and they are - $ 1 - $ Bromobutane and $ 2 - $ Bromobutane, $ 1 - $ Bromo - $ 2 - $ methylpropane, these three we just read above, but the fourth isomer that is possible is – tert-butyl bromide or $ 2 - $ Bromo - $ 2 - $ methylpropane. These bromobutane are less soluble in water and are denser than water.
Complete Step By Step Answer:
To solve this question we should have some insight regarding the conformation of structures.
Looking at our options - $ 1 - $ Bromobutane and $ 2 - $ Bromobutane, let’s have a look at their structure for the better understanding –


As, it is evident from the diagram above, that $ 1 - $ Bromobutane and $ 2 - $ Bromobutane fits completely into the formula $ {C_4}{H_9}Br $ . All the valencies of all the atoms are completely satisfied and have a stable structure under this formula - $ {C_4}{H_9}Br $ .
Similarly. $ 1 - $ Bromo - $ 2 - $ methyl propane – also has a stable structure satisfying all the valences of each atom.
But, coming to our option D , it is isobutane . It has a molecular formula of $ {C_4}{H_{10}} $ . Isobutane does not consist of bromine atoms. So it does not fit in our given formula $ {C_4}{H_9}Br $ .
So, the correct option to our question is D i.e. isobutane.
Note :
For $ {C_4}{H_9}Br $ we have four possible structural isomers and they are - $ 1 - $ Bromobutane and $ 2 - $ Bromobutane, $ 1 - $ Bromo - $ 2 - $ methylpropane, these three we just read above, but the fourth isomer that is possible is – tert-butyl bromide or $ 2 - $ Bromo - $ 2 - $ methylpropane. These bromobutane are less soluble in water and are denser than water.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




