Chemical formula of red lead is:
A. PbO
B. $\text{Pb}{{\text{O}}_{\text{2}}}$
C. $\text{P}{{\text{b}}_{3}}{{\text{O}}_{4}}$
D. $\text{P}{{\text{b}}_{2}}{{\text{O}}_{3}}$
Answer
615.6k+ views
Hint: We need to know the structure of red lead at first. Then we can count the number of oxygen and lead atoms in the compound.
Complete step by step answer:
Red lead is also known as minium. It is an organic compound and it has a bright reddish orange colour. Red lead is very useful in the manufacture of batteries, rust proof primer paints, etc. It is also an example of a mixed valence compound since it is composed of both lead (II) and lead (IV) in the ratio of 2:1. Lead (II)(IV) oxide has a tetragonal crystal structure at room temperature and it transforms to an orthorhombic at temperatures of 170 K or $-103{}^\circ C$. This translation of its phase, changes only the symmetry of the crystal and slightly modifies the interatomic distances and angles. This compound is prepared by the calcination of lead (II) oxide in air at about $\text{450 }\!\!{}^\circ\!\!\text{ C}$ to $\text{480 }\!\!{}^\circ\!\!\text{ C}$. The chemical reaction for the same is given below:
\[\text{6 }\!\!~\!\!\text{ PbO + }{{\text{O}}_{\text{2}}}\text{ }\!\!~\!\!\text{ }\xrightarrow{{}}\text{ 2P}{{\text{b}}_{\text{3}}}{{\text{O}}_{\text{4}}}\]
The chemical structure of red lead is given below from which we can count that there are 3 atoms of lead centred with 4 neighbouring atoms of oxygen hence forming the equation for red lead as $\text{P}{{\text{b}}_{3}}{{\text{O}}_{4}}$. Having a melting point of around $\text{500 }\!\!{}^\circ\!\!\text{ C}$, this compound exists in the form of vivid orange crystals.
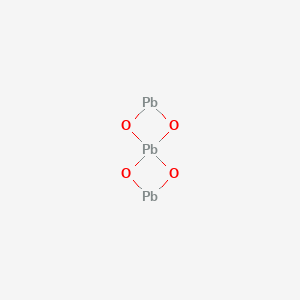
Therefore, the chemical formula for red lead is $\text{P}{{\text{b}}_{3}}{{\text{O}}_{4}}$
So, the correct answer is “Option C”.
Note: Natural red lead is very rare and it is formed only in extreme oxidizing conditions of lead ore bodies. The red lead is virtually insoluble in water and also in ethanol, though it is soluble in hydrochloric acid present in the stomach and therefore very toxic when ingested. It also dissolves in glacial acetic acid and diluted mixture of nitric acid and hydrogen peroxide also. When it is heated to a temperature of about $500{}^\circ C$ it decomposes to lead (II) oxide and oxygen, and at the temperature of $580{}^\circ C$, the reaction gets complete. The chemical equation for the same is given below:
\[\text{2P}{{\text{b}}_{\text{3}}}{{\text{O}}_{\text{4}}}\text{ }\!\!~\!\!\text{ }\xrightarrow{{}}\text{ 6PbO + }{{\text{O}}_{\text{2}}}\]
Complete step by step answer:
Red lead is also known as minium. It is an organic compound and it has a bright reddish orange colour. Red lead is very useful in the manufacture of batteries, rust proof primer paints, etc. It is also an example of a mixed valence compound since it is composed of both lead (II) and lead (IV) in the ratio of 2:1. Lead (II)(IV) oxide has a tetragonal crystal structure at room temperature and it transforms to an orthorhombic at temperatures of 170 K or $-103{}^\circ C$. This translation of its phase, changes only the symmetry of the crystal and slightly modifies the interatomic distances and angles. This compound is prepared by the calcination of lead (II) oxide in air at about $\text{450 }\!\!{}^\circ\!\!\text{ C}$ to $\text{480 }\!\!{}^\circ\!\!\text{ C}$. The chemical reaction for the same is given below:
\[\text{6 }\!\!~\!\!\text{ PbO + }{{\text{O}}_{\text{2}}}\text{ }\!\!~\!\!\text{ }\xrightarrow{{}}\text{ 2P}{{\text{b}}_{\text{3}}}{{\text{O}}_{\text{4}}}\]
The chemical structure of red lead is given below from which we can count that there are 3 atoms of lead centred with 4 neighbouring atoms of oxygen hence forming the equation for red lead as $\text{P}{{\text{b}}_{3}}{{\text{O}}_{4}}$. Having a melting point of around $\text{500 }\!\!{}^\circ\!\!\text{ C}$, this compound exists in the form of vivid orange crystals.

Therefore, the chemical formula for red lead is $\text{P}{{\text{b}}_{3}}{{\text{O}}_{4}}$
So, the correct answer is “Option C”.
Note: Natural red lead is very rare and it is formed only in extreme oxidizing conditions of lead ore bodies. The red lead is virtually insoluble in water and also in ethanol, though it is soluble in hydrochloric acid present in the stomach and therefore very toxic when ingested. It also dissolves in glacial acetic acid and diluted mixture of nitric acid and hydrogen peroxide also. When it is heated to a temperature of about $500{}^\circ C$ it decomposes to lead (II) oxide and oxygen, and at the temperature of $580{}^\circ C$, the reaction gets complete. The chemical equation for the same is given below:
\[\text{2P}{{\text{b}}_{\text{3}}}{{\text{O}}_{\text{4}}}\text{ }\!\!~\!\!\text{ }\xrightarrow{{}}\text{ 6PbO + }{{\text{O}}_{\text{2}}}\]
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life




