Bond order of \[\text{B}{{\text{e}}_{\text{2}}}\] is
A. 1
B. 2
C. 3
D. 0
Answer
598.5k+ views
Hint: According to the molecular orbital theory, the bond order is defined as the number of covalent bonds in a molecule. Bond order is equal to half of the difference between the number of electrons in bonding (\[{{N}_{b}}\]) and antibonding molecular orbitals (\[{{N}_{a}}\]).
Complete Solution :
\[\text{B}{{\text{e}}_{\text{2}}}\] molecule will be formed by the overlapping of atomic orbitals of two beryllium atoms.
A Be atom has four electrons. It has two valence electrons and its electronic configuration is \[1{{s}^{2}}2{{s}^{2}}\]. Therefore, \[\text{B}{{\text{e}}_{\text{2}}}\] molecule has eight electrons which are to be filled in four molecular orbitals.
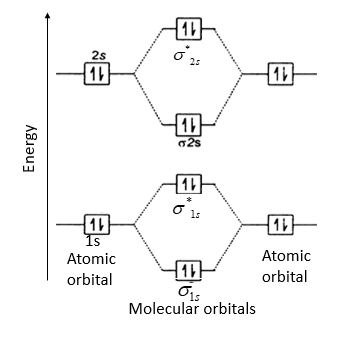
Thus, electronic configuration of \[\text{B}{{\text{e}}_{\text{2}}}\] is \[{{\left( \sigma 1s \right)}^{2}}{{\left( {{\sigma }^{*}}1s \right)}^{2}}{{\left( \sigma 2s \right)}^{2}}{{\left( {{\sigma }^{*}}2s \right)}^{2}}\]
Here, bonding electrons, \[{{N}_{b}}\] = 4 and anti-bonding electrons, \[{{N}_{a}}\]= 4
Therefore, bond order (B.O.) of \[\text{B}{{\text{e}}_{\text{2}}}\] molecule is
\[\begin{align}
& \text{B}\text{.O}\text{.=}\frac{1}{2}({{N}_{b}}-{{N}_{a}}) \\
& \text{B}\text{.O}\text{.}=\frac{1}{2}(4-4)=0 \\
\end{align}\]
- Zero value of bond order corresponds to non-existence of \[\text{B}{{\text{e}}_{\text{2}}}\] molecule.
So, the correct answer is “Option D”.
Note: The bond order of a molecule conveys the following information:
1. The stability of a molecule can also be expressed in terms of bond order. Higher the bond order, more stable is the molecule.
2. Bond length: Bond order and bond length are inversely related. Thus, higher the bond order, shorter is the bond length and vice-versa.
3. Bond dissociation energy: Bond order in a molecule is directly proportional to its bond dissociation energy. Greater the bond order, more will be the value of bond dissociation energy.
Complete Solution :
\[\text{B}{{\text{e}}_{\text{2}}}\] molecule will be formed by the overlapping of atomic orbitals of two beryllium atoms.
A Be atom has four electrons. It has two valence electrons and its electronic configuration is \[1{{s}^{2}}2{{s}^{2}}\]. Therefore, \[\text{B}{{\text{e}}_{\text{2}}}\] molecule has eight electrons which are to be filled in four molecular orbitals.

Thus, electronic configuration of \[\text{B}{{\text{e}}_{\text{2}}}\] is \[{{\left( \sigma 1s \right)}^{2}}{{\left( {{\sigma }^{*}}1s \right)}^{2}}{{\left( \sigma 2s \right)}^{2}}{{\left( {{\sigma }^{*}}2s \right)}^{2}}\]
Here, bonding electrons, \[{{N}_{b}}\] = 4 and anti-bonding electrons, \[{{N}_{a}}\]= 4
Therefore, bond order (B.O.) of \[\text{B}{{\text{e}}_{\text{2}}}\] molecule is
\[\begin{align}
& \text{B}\text{.O}\text{.=}\frac{1}{2}({{N}_{b}}-{{N}_{a}}) \\
& \text{B}\text{.O}\text{.}=\frac{1}{2}(4-4)=0 \\
\end{align}\]
- Zero value of bond order corresponds to non-existence of \[\text{B}{{\text{e}}_{\text{2}}}\] molecule.
So, the correct answer is “Option D”.
Note: The bond order of a molecule conveys the following information:
1. The stability of a molecule can also be expressed in terms of bond order. Higher the bond order, more stable is the molecule.
2. Bond length: Bond order and bond length are inversely related. Thus, higher the bond order, shorter is the bond length and vice-versa.
3. Bond dissociation energy: Bond order in a molecule is directly proportional to its bond dissociation energy. Greater the bond order, more will be the value of bond dissociation energy.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Explain zero factorial class 11 maths CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




