What is the bond angle in a tetrahedral molecule?
Answer
530.7k+ views
Hint: According to the VSEPR theory, each atom in a molecule will obtain a shape that minimises repulsion between electrons in that atom's valence shell. It's essentially a model for predicting molecular geometry. The bonding and molecular geometry of organic molecules and polyatomic ions are the main focus of VSEPR models.
Complete answer:
As we know, tetra means four and hedral means a solid face, therefore tetrahedral means "having four faces." When four bonds are all linked to one core atom and there are no lone electron pairs, this shape is formed.
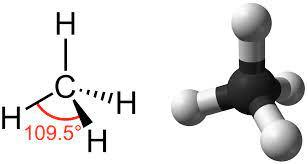
A tetrahedral molecule is made up $ 4 $ equally spaced $ s{p^3} $ hybrid orbitals forming bond angles of $ {109.5^ \circ } $
Example of a tetrahedral molecule is methane.
The four equivalent bonds ( $ C - H $ ) point in four geometrically equivalent directions in three dimensions which can be corresponded to the four corners of a tetrahedron as hydrogen, centred on the carbon atom.
Additional Information:
The VSEPR model accurately predicts the $ 3 $ -D shape of molecules and ions, but it fails to provide any detailed information on bond length or bond structure.
VSEPR models are based on the idea that electrons revolving around a core atom will arrange themselves to minimise repulsion, dictating the molecule's shape.
It can predict the form of almost every compound with a centre atom.
Note:
When counting bond angles, always use shapes above geometries of the given molecule because bond angles are always between bonds, not between lone pairs. Shape primarily examines real bonds, whereas geometry considers the total arrangement of a molecule, including bonds and lone pairs.
Complete answer:
As we know, tetra means four and hedral means a solid face, therefore tetrahedral means "having four faces." When four bonds are all linked to one core atom and there are no lone electron pairs, this shape is formed.
A tetrahedral molecule is made up $ 4 $ equally spaced $ s{p^3} $ hybrid orbitals forming bond angles of $ {109.5^ \circ } $
Example of a tetrahedral molecule is methane.
The four equivalent bonds ( $ C - H $ ) point in four geometrically equivalent directions in three dimensions which can be corresponded to the four corners of a tetrahedron as hydrogen, centred on the carbon atom.

Additional Information:
The VSEPR model accurately predicts the $ 3 $ -D shape of molecules and ions, but it fails to provide any detailed information on bond length or bond structure.
VSEPR models are based on the idea that electrons revolving around a core atom will arrange themselves to minimise repulsion, dictating the molecule's shape.
It can predict the form of almost every compound with a centre atom.
Note:
When counting bond angles, always use shapes above geometries of the given molecule because bond angles are always between bonds, not between lone pairs. Shape primarily examines real bonds, whereas geometry considers the total arrangement of a molecule, including bonds and lone pairs.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




