Benzonitrile is the IUPAC name of which compound?
Answer
538.2k+ views
Hint: We need to know that the organic compound that contains a cyano functional group is known as a Nitrile. Denoted by $ - C{N^ - }$. In this the carbon and nitrogen gas a triple bond i.e. $ - C \equiv N$. The general chemical formula of nitrile compound is RCN, where R is alkyl/aryl group. The common name for benzonitrile is Cyanobenzene or phenyl cyanide. Benzonitrile is slightly soluble in water.
Complete answer:
We need to know that the benzonitrile consists of a benzene ring and a nitrile group and has a chemical formula of ${C_6}{H_5} - CN$ or $Ph - CN$. This compound is colourless with a sweet bitter almond odour.
The molecule of Benzonitrile contains $13$ bonds in total; $8$ non- H bonds, $7$ multiple bonds, $1$ triple bond, $6$ aromatic bonds, $1$ six membered ring and 1 nitrile. The structure of Benzonitrile is as shown below:
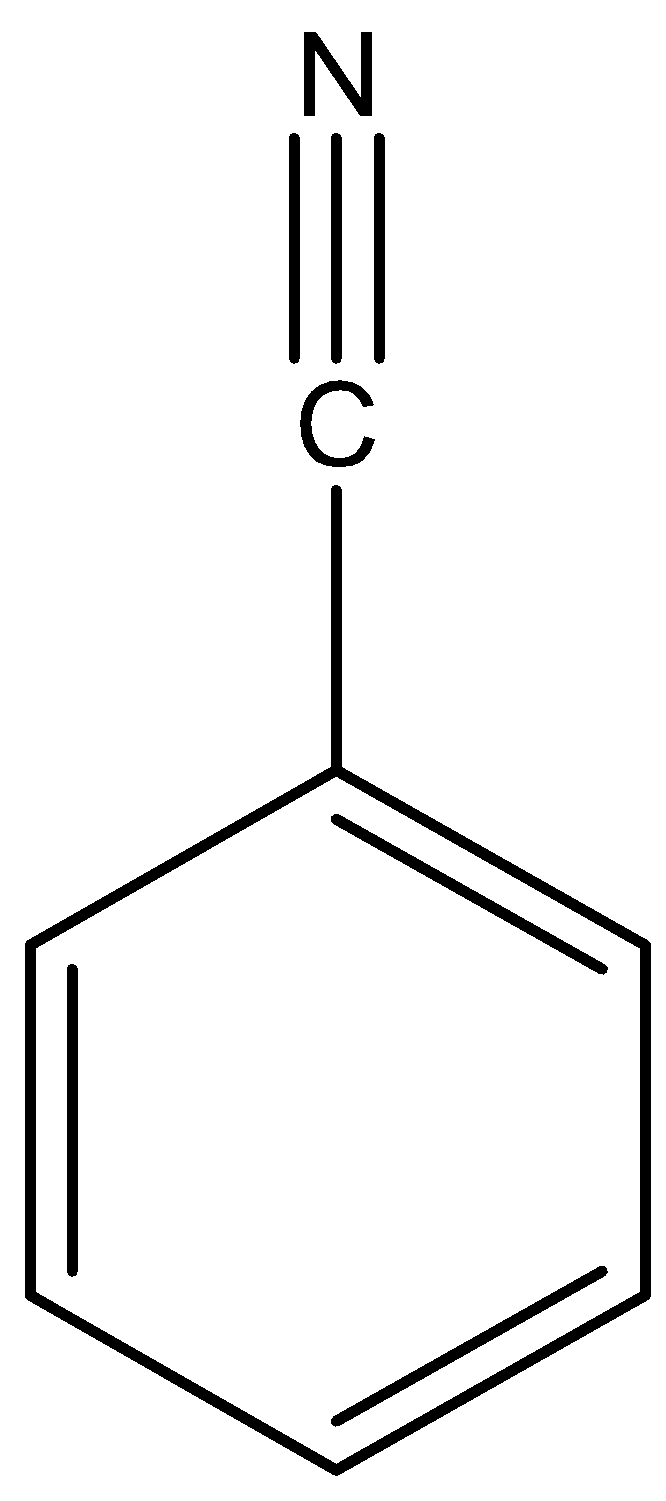
Benzonitrile is a useful solvent and a precursor for many derivatives. It is a precursor for Diphenyl Ketamine. Benzonitrile also forms coordination compounds with transition metal elements that are soluble in organic solvents. The benzonitrile ligand can be readily displaced by stronger ligands.
Additional Information:
We need to know that Benzonitrile is widely used in the rubber industry as a rubber chemical and also as a solvent for nitrile rubber, resins, and polymers and also for many anhydrous salts. It is primarily a precursor for benzoguanamine.
Note:
We have to remember that benzonitrile is produced by ammoxidation of toluene. It is the reaction with ammonia and oxygen at \[400 - 450^\circ C\]. In the laboratory it is prepared by the dehydration of benzamide or by Rosenmund-von Braun reaction using $CuCN$ or $NaCN$ and bromobenzene.
Complete answer:
We need to know that the benzonitrile consists of a benzene ring and a nitrile group and has a chemical formula of ${C_6}{H_5} - CN$ or $Ph - CN$. This compound is colourless with a sweet bitter almond odour.
The molecule of Benzonitrile contains $13$ bonds in total; $8$ non- H bonds, $7$ multiple bonds, $1$ triple bond, $6$ aromatic bonds, $1$ six membered ring and 1 nitrile. The structure of Benzonitrile is as shown below:

Benzonitrile is a useful solvent and a precursor for many derivatives. It is a precursor for Diphenyl Ketamine. Benzonitrile also forms coordination compounds with transition metal elements that are soluble in organic solvents. The benzonitrile ligand can be readily displaced by stronger ligands.
Additional Information:
We need to know that Benzonitrile is widely used in the rubber industry as a rubber chemical and also as a solvent for nitrile rubber, resins, and polymers and also for many anhydrous salts. It is primarily a precursor for benzoguanamine.
Note:
We have to remember that benzonitrile is produced by ammoxidation of toluene. It is the reaction with ammonia and oxygen at \[400 - 450^\circ C\]. In the laboratory it is prepared by the dehydration of benzamide or by Rosenmund-von Braun reaction using $CuCN$ or $NaCN$ and bromobenzene.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




