Benzene diazonium chloride on reaction with phenol in a basic medium gives:
A) diphenyl ether
B) p-Hydroxy azobenzene
C) chlorobenzene
D) benzene
Answer
627k+ views
Hint: When the concentration of OH- ion in solution is more, it is basic in nature. We call such a solution a basic medium.
Complete step by step answer:
Benzene diazonium chloride in reaction with phenol in a basic medium gives p-Hydroxy azobenzene.
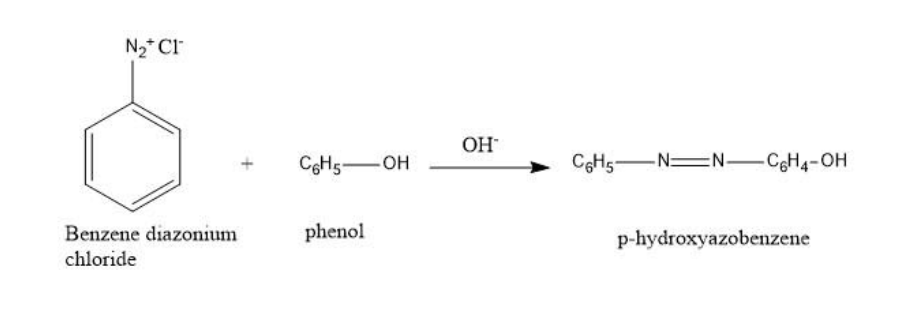
So, out of the given options, B is the correct option, that is, p-Hydroxy azobenzene.
Additional information:
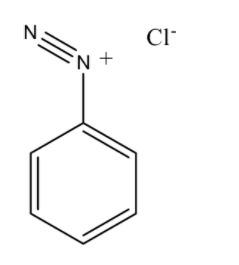
The structure of Benzene diazonium chloride is
And the structure of p-Hydroxy azobenzene is,
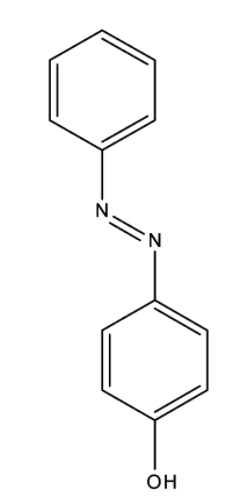
Benzene diazonium chloride has the chemical formula ${{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{5}}}{\rm{Cl}}{{\rm{N}}_{\rm{2}}}$. Benzene diazonium chloride is a salt of a chloride and diazonium cation. Benzene diazonium chloride exists as a colourless solid and it is soluble in polar solvents such as in water. It acts as the parent member of the aryl diazonium compounds. This compound is widely used in organic chemistry. As this salt is unstable, it is not available commercially but is prepared according to the demand. p-hydroxyazobenzene has chemical formula ${\rm{C}}{{\rm{l}}_{\rm{2}}}{{\rm{H}}_{{\rm{10}}}}{{\rm{N}}_{{\rm{20}}}}$ and has the atomic number 198.221. It has a density of $1.13{\rm{g/c}}{{\rm{m}}^{\rm{3}}}$, and has a boiling point of ${369.812^o}C$ and has a refractive index of 1.596.
The mechanism for the reaction of benzene diazonium chloride on reaction with phenol can be given as follows:
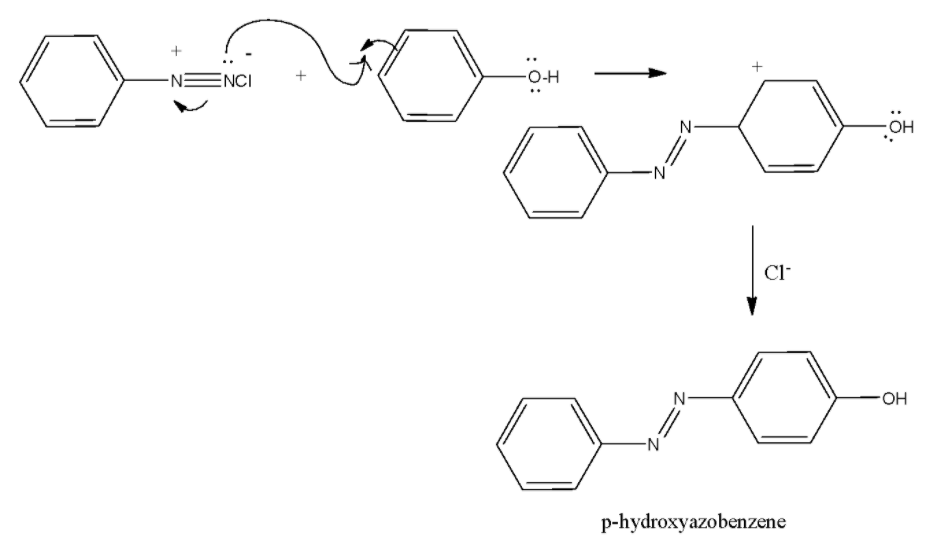
Note:
Benzene diazonium chloride has the chemical formula ${{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{5}}}{\rm{Cl}}{{\rm{N}}_{\rm{2}}}$. This compound is soluble in polar solvent and it exists as a colourless solid.
Complete step by step answer:
Benzene diazonium chloride in reaction with phenol in a basic medium gives p-Hydroxy azobenzene.

So, out of the given options, B is the correct option, that is, p-Hydroxy azobenzene.
Additional information:
The structure of Benzene diazonium chloride is

And the structure of p-Hydroxy azobenzene is,

Benzene diazonium chloride has the chemical formula ${{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{5}}}{\rm{Cl}}{{\rm{N}}_{\rm{2}}}$. Benzene diazonium chloride is a salt of a chloride and diazonium cation. Benzene diazonium chloride exists as a colourless solid and it is soluble in polar solvents such as in water. It acts as the parent member of the aryl diazonium compounds. This compound is widely used in organic chemistry. As this salt is unstable, it is not available commercially but is prepared according to the demand. p-hydroxyazobenzene has chemical formula ${\rm{C}}{{\rm{l}}_{\rm{2}}}{{\rm{H}}_{{\rm{10}}}}{{\rm{N}}_{{\rm{20}}}}$ and has the atomic number 198.221. It has a density of $1.13{\rm{g/c}}{{\rm{m}}^{\rm{3}}}$, and has a boiling point of ${369.812^o}C$ and has a refractive index of 1.596.
The mechanism for the reaction of benzene diazonium chloride on reaction with phenol can be given as follows:

Note:
Benzene diazonium chloride has the chemical formula ${{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{5}}}{\rm{Cl}}{{\rm{N}}_{\rm{2}}}$. This compound is soluble in polar solvent and it exists as a colourless solid.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




