Assuming 2s-2p mixing is not operative the paramagnetic species among the following is:
A) $\text{ B}{{\text{e}}_{\text{2 }}}$
B) $\text{ }{{\text{N}}_{\text{2 }}}$
C) $\text{ }{{\text{C}}_{\text{2 }}}$
D) $\text{ }{{\text{B}}_{\text{2 }}}$
Answer
589.8k+ views
Hint: The magnetic property of a molecule can be explained based on the molecular orbital theory. The molecule which does not contain the unpaired electron is known as the paramagnetic. The molecule which has all-electron paid-up does not contribute towards the magnetic property. It is diamagnetic in nature. To solve such a problem write down the MOT diagram of molecules.
Complete step by step solution:
The MOT diagram is drawn in such a way that we do not consider the s and p orbital mixing. Therefore the order of orbitals is not changed.
Let’s first draw the MOT of all molecules given in the problem.
A) $\text{ B}{{\text{e}}_{\text{2 }}}$ molecule: Beryllium atom has two electrons in the first shell and two-electrons in the second shell. The electronic configuration of the beryllium atom is as shown below,
$\text{ Be = 1}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{s}}^{\text{2}}}\text{ }$
The $\text{ B}{{\text{e}}_{\text{2 }}}$molecule contains a total of 8 electrons in it. These 8 electrons are arranged in the molecular orbitals.
The MOT is as shown below,
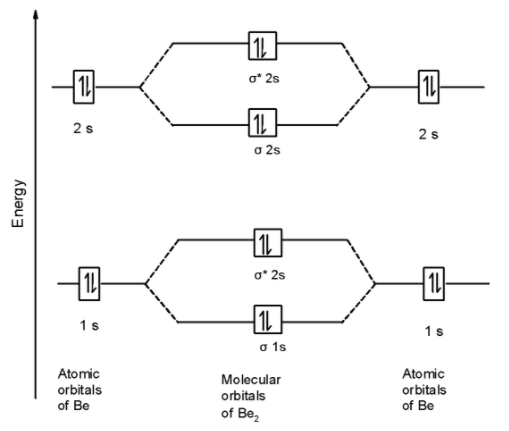
First of all, we can write the molecular orbital configuration of $\text{ B}{{\text{e}}_{\text{2}}}$ . The molecular orbital configuration of $\text{ B}{{\text{e}}_{\text{2}}}$ the molecule is as follows:
\[\]\[\text{ }\!\!\sigma\!\!\text{ 1}{{\text{s}}^{\text{2}}}\text{,}{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{1}{{\text{s}}^{\text{2}}}\text{, }\!\!\sigma\!\!\text{ 2}{{\text{s}}^{\text{2}}}\text{, }{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{2}{{\text{s}}^{\text{2}}}\text{ OR KK , }\!\!\sigma\!\!\text{ 2}{{\text{s}}^{\text{2}}}\text{, }{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{2}{{\text{s}}^{\text{2}}}\text{ }\]
Since, here all molecular orbitals contain the two electrons. These are all paired electrons. Thus $\text{ B}{{\text{e}}_{\text{2}}}$ the molecule is diamagnetic.
B) $\text{ }{{\text{N}}_{\text{2 }}}$ molecule : The nitrogen atom has three electrons in the 2p shell and 4 electrons in the innermost shell. The electronic configuration of nitrogen atom is as shown below,
$\text{ N = 1}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{p}}^{\text{1}}}_{\text{x}}\text{= 2}{{\text{p}}^{\text{1}}}_{\text{y}}=\text{2}{{\text{p}}^{\text{1}}}_{z}\text{ }$
The $\text{ }{{\text{N}}_{\text{2 }}}$molecule contains a total of 14 electrons in it. These 14 electrons are arranged in the molecular orbitals. The MOT is as shown below,
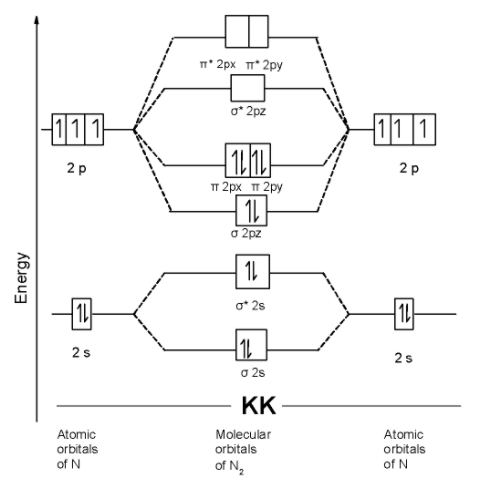
We can write the molecular orbital configuration of $\text{ }{{\text{N}}_{\text{2 }}}$ . The molecular orbital configuration of $\text{ B}{{\text{e}}_{\text{2}}}$ the molecule is as follows:
$\text{ }\!\!\sigma\!\!\text{ 1}{{\text{s}}^{\text{2}}}\text{,}{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{1}{{\text{s}}^{\text{2}}}\text{, }\!\!\sigma\!\!\text{ 2}{{\text{s}}^{\text{2}}}\text{, }{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{2}{{\text{s}}^{\text{2}}}\text{ , }\!\!\pi\!\!\text{ 2p}_{\text{y}}^{\text{2}}\text{ = }\!\!\pi\!\!\text{ 2p}_{\text{x}}^{\text{2}}\text{ , }\!\!\sigma\!\!\text{ 2p}_{\text{z}}^{\text{2}}\text{ }$
Since, here all molecular orbitals contain the two electrons. These are all paired electrons. Thus $\text{ }{{\text{N}}_{\text{2 }}}$ the molecule is diamagnetic.
C) $\text{ }{{\text{C}}_{\text{2 }}}$ Molecule: The carbon atom has two electrons in the 2p shell and 4 electrons in the innermost shell. The electronic configuration of nitrogen atom is as shown below,$\text{ C = 1}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{p}}^{\text{1}}}_{\text{x}}\text{= 2}{{\text{p}}^{\text{1}}}_{\text{y}}=\text{2}{{\text{p}}^{0}}_{z}\text{ }$
The $\text{ }{{\text{C}}_{\text{2 }}}$molecule contains a total of 12 electrons in it. These 12 electrons are arranged in the molecular orbitals.
The MOT is as shown below,
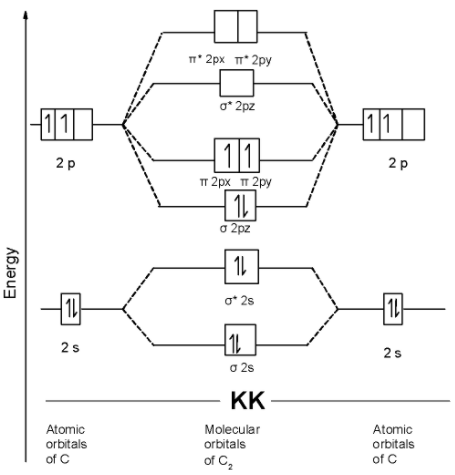
we can write the molecular orbital configuration of $\text{ }{{\text{C}}_{\text{2 }}}$ . The molecular orbital configuration of $\text{ }{{\text{C}}_{\text{2}}}$ the molecule is as follows:
$\text{ }\!\!\sigma\!\!\text{ 1}{{\text{s}}^{\text{2}}}\text{,}{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{1}{{\text{s}}^{\text{2}}}\text{, }\!\!\sigma\!\!\text{ 2}{{\text{s}}^{\text{2}}}\text{, }{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{2}{{\text{s}}^{\text{2}}}\text{ , }\!\!\sigma\!\!\text{ 2p}_{\text{z}}^{\text{2}},\text{ }\!\!\pi\!\!\text{ 2p}_{\text{y}}^{1}\text{ = }\!\!\pi\!\!\text{ 2p}_{\text{x}}^{1}\text{ }$
Since the pi molecular orbitals contain unpaired electrons. Thus $\text{ }{{\text{C}}_{\text{2 }}}$ the molecule is paramagnetic.
D) $\text{ }{{\text{B}}_{\text{2 }}}$molecule: The boron atom has one electron in the 2p shell and 4 electrons in the innermost shell. The electronic configuration of nitrogen atom is as shown below,
$\text{ B = 1}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{p}}^{\text{1}}}_{\text{x}}\text{= 2}{{\text{p}}^{0}}_{\text{y}}=\text{2}{{\text{p}}^{0}}_{z}\text{ }$
The $\text{ }{{\text{B}}_{\text{2 }}}$molecule contains a total of 10 electrons in it. These 10 electrons are arranged in the molecular orbitals.
The MOT is as shown below,
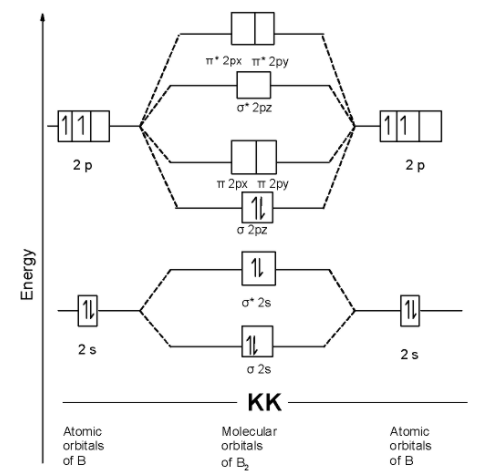
We can write the molecular orbital configuration of $\text{ }{{\text{B}}_{\text{2 }}}$ . The molecular orbital configuration of $\text{ }{{\text{B}}_{\text{2 }}}$ the molecule is as follows:$\text{ }\!\!\sigma\!\!\text{ 1}{{\text{s}}^{\text{2}}}\text{,}{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{1}{{\text{s}}^{\text{2}}}\text{, }\!\!\sigma\!\!\text{ 2}{{\text{s}}^{\text{2}}}\text{, }{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{2}{{\text{s}}^{\text{2}}}\text{ , }\!\!\sigma\!\!\text{ 2p}_{\text{z}}^{\text{2}}\text{ }$
Since the molecular orbitals theory does not contain the unpaired electron. Thus $\text{ }{{\text{B}}_{\text{2 }}}$ the molecule is diamagnetic.
Thus, from here we know that only $\text{ }{{\text{C}}_{\text{2 }}}$ molecule is paramagnetic.
Hence, (C) is the correct option.
Note: Note that, in atoms like $\text{ Li }$ , $\text{ Be }$ , $\text{ C }$ , $\text{ N }$ the energy difference is very small between the $\text{ 2s }$ and $\text{ 2p }$ orbitals. These are close to each other and thus p and s orbitals combine and lead to change in the expected order of the orbital energies. Here, we are not considering the mixing of s and p orbitals thus the carbon molecule is paramagnetic otherwise if we consider the mixing then the carbon molecule is diamagnetic.
Complete step by step solution:
The MOT diagram is drawn in such a way that we do not consider the s and p orbital mixing. Therefore the order of orbitals is not changed.
Let’s first draw the MOT of all molecules given in the problem.
A) $\text{ B}{{\text{e}}_{\text{2 }}}$ molecule: Beryllium atom has two electrons in the first shell and two-electrons in the second shell. The electronic configuration of the beryllium atom is as shown below,
$\text{ Be = 1}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{s}}^{\text{2}}}\text{ }$
The $\text{ B}{{\text{e}}_{\text{2 }}}$molecule contains a total of 8 electrons in it. These 8 electrons are arranged in the molecular orbitals.
The MOT is as shown below,

First of all, we can write the molecular orbital configuration of $\text{ B}{{\text{e}}_{\text{2}}}$ . The molecular orbital configuration of $\text{ B}{{\text{e}}_{\text{2}}}$ the molecule is as follows:
\[\]\[\text{ }\!\!\sigma\!\!\text{ 1}{{\text{s}}^{\text{2}}}\text{,}{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{1}{{\text{s}}^{\text{2}}}\text{, }\!\!\sigma\!\!\text{ 2}{{\text{s}}^{\text{2}}}\text{, }{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{2}{{\text{s}}^{\text{2}}}\text{ OR KK , }\!\!\sigma\!\!\text{ 2}{{\text{s}}^{\text{2}}}\text{, }{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{2}{{\text{s}}^{\text{2}}}\text{ }\]
Since, here all molecular orbitals contain the two electrons. These are all paired electrons. Thus $\text{ B}{{\text{e}}_{\text{2}}}$ the molecule is diamagnetic.
B) $\text{ }{{\text{N}}_{\text{2 }}}$ molecule : The nitrogen atom has three electrons in the 2p shell and 4 electrons in the innermost shell. The electronic configuration of nitrogen atom is as shown below,
$\text{ N = 1}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{p}}^{\text{1}}}_{\text{x}}\text{= 2}{{\text{p}}^{\text{1}}}_{\text{y}}=\text{2}{{\text{p}}^{\text{1}}}_{z}\text{ }$
The $\text{ }{{\text{N}}_{\text{2 }}}$molecule contains a total of 14 electrons in it. These 14 electrons are arranged in the molecular orbitals. The MOT is as shown below,

We can write the molecular orbital configuration of $\text{ }{{\text{N}}_{\text{2 }}}$ . The molecular orbital configuration of $\text{ B}{{\text{e}}_{\text{2}}}$ the molecule is as follows:
$\text{ }\!\!\sigma\!\!\text{ 1}{{\text{s}}^{\text{2}}}\text{,}{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{1}{{\text{s}}^{\text{2}}}\text{, }\!\!\sigma\!\!\text{ 2}{{\text{s}}^{\text{2}}}\text{, }{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{2}{{\text{s}}^{\text{2}}}\text{ , }\!\!\pi\!\!\text{ 2p}_{\text{y}}^{\text{2}}\text{ = }\!\!\pi\!\!\text{ 2p}_{\text{x}}^{\text{2}}\text{ , }\!\!\sigma\!\!\text{ 2p}_{\text{z}}^{\text{2}}\text{ }$
Since, here all molecular orbitals contain the two electrons. These are all paired electrons. Thus $\text{ }{{\text{N}}_{\text{2 }}}$ the molecule is diamagnetic.
C) $\text{ }{{\text{C}}_{\text{2 }}}$ Molecule: The carbon atom has two electrons in the 2p shell and 4 electrons in the innermost shell. The electronic configuration of nitrogen atom is as shown below,$\text{ C = 1}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{p}}^{\text{1}}}_{\text{x}}\text{= 2}{{\text{p}}^{\text{1}}}_{\text{y}}=\text{2}{{\text{p}}^{0}}_{z}\text{ }$
The $\text{ }{{\text{C}}_{\text{2 }}}$molecule contains a total of 12 electrons in it. These 12 electrons are arranged in the molecular orbitals.
The MOT is as shown below,

we can write the molecular orbital configuration of $\text{ }{{\text{C}}_{\text{2 }}}$ . The molecular orbital configuration of $\text{ }{{\text{C}}_{\text{2}}}$ the molecule is as follows:
$\text{ }\!\!\sigma\!\!\text{ 1}{{\text{s}}^{\text{2}}}\text{,}{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{1}{{\text{s}}^{\text{2}}}\text{, }\!\!\sigma\!\!\text{ 2}{{\text{s}}^{\text{2}}}\text{, }{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{2}{{\text{s}}^{\text{2}}}\text{ , }\!\!\sigma\!\!\text{ 2p}_{\text{z}}^{\text{2}},\text{ }\!\!\pi\!\!\text{ 2p}_{\text{y}}^{1}\text{ = }\!\!\pi\!\!\text{ 2p}_{\text{x}}^{1}\text{ }$
Since the pi molecular orbitals contain unpaired electrons. Thus $\text{ }{{\text{C}}_{\text{2 }}}$ the molecule is paramagnetic.
D) $\text{ }{{\text{B}}_{\text{2 }}}$molecule: The boron atom has one electron in the 2p shell and 4 electrons in the innermost shell. The electronic configuration of nitrogen atom is as shown below,
$\text{ B = 1}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{s}}^{\text{2}}}\text{ 2}{{\text{p}}^{\text{1}}}_{\text{x}}\text{= 2}{{\text{p}}^{0}}_{\text{y}}=\text{2}{{\text{p}}^{0}}_{z}\text{ }$
The $\text{ }{{\text{B}}_{\text{2 }}}$molecule contains a total of 10 electrons in it. These 10 electrons are arranged in the molecular orbitals.
The MOT is as shown below,

We can write the molecular orbital configuration of $\text{ }{{\text{B}}_{\text{2 }}}$ . The molecular orbital configuration of $\text{ }{{\text{B}}_{\text{2 }}}$ the molecule is as follows:$\text{ }\!\!\sigma\!\!\text{ 1}{{\text{s}}^{\text{2}}}\text{,}{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{1}{{\text{s}}^{\text{2}}}\text{, }\!\!\sigma\!\!\text{ 2}{{\text{s}}^{\text{2}}}\text{, }{{\text{ }\!\!\sigma\!\!\text{ }}^{\text{*}}}\text{2}{{\text{s}}^{\text{2}}}\text{ , }\!\!\sigma\!\!\text{ 2p}_{\text{z}}^{\text{2}}\text{ }$
Since the molecular orbitals theory does not contain the unpaired electron. Thus $\text{ }{{\text{B}}_{\text{2 }}}$ the molecule is diamagnetic.
Thus, from here we know that only $\text{ }{{\text{C}}_{\text{2 }}}$ molecule is paramagnetic.
Hence, (C) is the correct option.
Note: Note that, in atoms like $\text{ Li }$ , $\text{ Be }$ , $\text{ C }$ , $\text{ N }$ the energy difference is very small between the $\text{ 2s }$ and $\text{ 2p }$ orbitals. These are close to each other and thus p and s orbitals combine and lead to change in the expected order of the orbital energies. Here, we are not considering the mixing of s and p orbitals thus the carbon molecule is paramagnetic otherwise if we consider the mixing then the carbon molecule is diamagnetic.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Discuss the various forms of bacteria class 11 biology CBSE




