Arrhenius equation is $k=A{{e}^{\dfrac{-{{E}_{a}}}{RT}}}$. Which of the following graphs represents the variation of rate constant k against the temperature T?
(A)
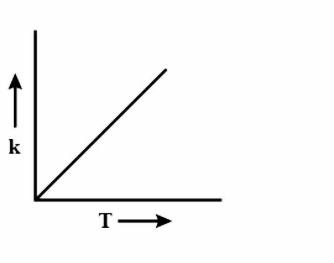
(B)
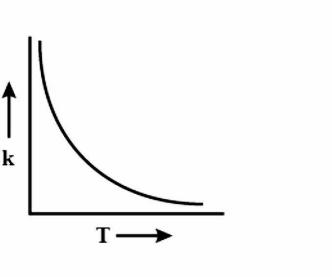
(C)
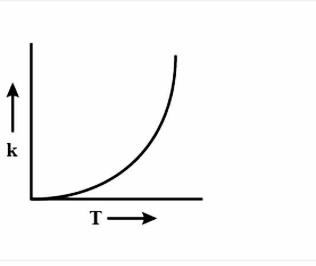
(D)
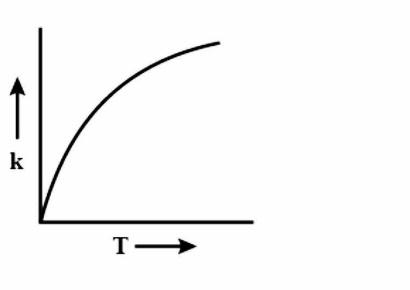




Answer
611.7k+ views
Note: You must recall the fact that when the temperature is increased in a chemical reaction, then the rate constant also increases. For every \[{{10}^{0}}\] rise in temperature, the rate constant gets nearly doubled.
Complete Step-by-step solution:
First, let us recall the Arrhenius Equation for chemical kinetics. The Arrhenius equation is represented as:
$k=A{{e}^{\dfrac{-{{E}_{a}}}{RT}}}$ , k is the rate constant,
A is the Arrhenius factor,
${{E}_{a}}$ is the activation energy.
During every chemical reaction, an intermediate is formed, in the beginning. In order for the intermediate to form, some energy is required and this energy is called the Activation energy. A lot of collisions happen between the molecules during the reaction and these collisions are responsible for formation of the products. Now, if the temperature is increased, then heat is transferred in the form of Kinetic energy. The fraction of molecules which collide increases, which have energy more than the activation energy \[{{E}_{a}}\]. As the graph is increasing, so accordingly the options are eliminated. Now to figure out the shape of the graph, we have to again look at the equation i.e $k=A{{e}^{\dfrac{-{{E}_{a}}}{RT}}}$. Clearly, the equation is an exponential graph so it will be curved and not be in a straight line. Hence, it is either B or D. The last thing we can look at is the sign of the exponent.
We can observe that it is a negative exponential power graph, so in that case, our answer will be option C, i.e negative exponential.
Note: No chemical reaction can occur if the fraction of molecules have energies less than the activation energy \[{{E}_{a}}\]. Moreover, you can put random values of k and \[{{E}_{a}}\], A in the equation and check which graph satisfies it, in case time is less during the exam.
Complete Step-by-step solution:
First, let us recall the Arrhenius Equation for chemical kinetics. The Arrhenius equation is represented as:
$k=A{{e}^{\dfrac{-{{E}_{a}}}{RT}}}$ , k is the rate constant,
A is the Arrhenius factor,
${{E}_{a}}$ is the activation energy.
During every chemical reaction, an intermediate is formed, in the beginning. In order for the intermediate to form, some energy is required and this energy is called the Activation energy. A lot of collisions happen between the molecules during the reaction and these collisions are responsible for formation of the products. Now, if the temperature is increased, then heat is transferred in the form of Kinetic energy. The fraction of molecules which collide increases, which have energy more than the activation energy \[{{E}_{a}}\]. As the graph is increasing, so accordingly the options are eliminated. Now to figure out the shape of the graph, we have to again look at the equation i.e $k=A{{e}^{\dfrac{-{{E}_{a}}}{RT}}}$. Clearly, the equation is an exponential graph so it will be curved and not be in a straight line. Hence, it is either B or D. The last thing we can look at is the sign of the exponent.
We can observe that it is a negative exponential power graph, so in that case, our answer will be option C, i.e negative exponential.
Note: No chemical reaction can occur if the fraction of molecules have energies less than the activation energy \[{{E}_{a}}\]. Moreover, you can put random values of k and \[{{E}_{a}}\], A in the equation and check which graph satisfies it, in case time is less during the exam.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




