What are two conformations of cis-1,4-dimethyl cyclohexane?
Answer
558k+ views
Hint : Just two stereoisomers, cis- and trans- 1,4-dimethyl cyclohexane, have an internal symmetry plane. As a result, both are meso compounds (optically inactive). Only cis-1,3-dimethyl cyclohexane, unlike 1,4-dimethyl cyclohexane, has an internal symmetry plane, whereas trans-1,3-dimethyl cyclohexane does not.
Complete Step By Step Answer:
Cis-1,4-dimethyl cyclohexane has one chair conformation and two boat conformations. The chair conformation of cyclohexane is more stable than the boat conformation since the $C - H$ bonds are similarly axial and equatorial in the chair conformation, i.e., six of the twelve $C - H$bonds are axial and six are equatorial, and each carbon has one axial and one equatorial $C - H$bond.
The chain structure of cis-1,4-dimethyl cyclohexane
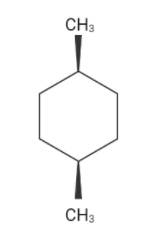
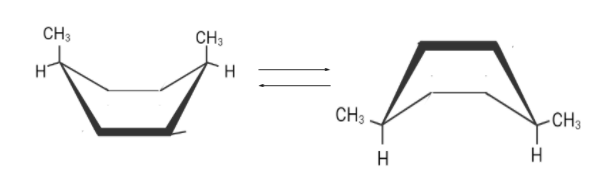
There are two flipped boat conformations . Although both are different but the right one is more stable.
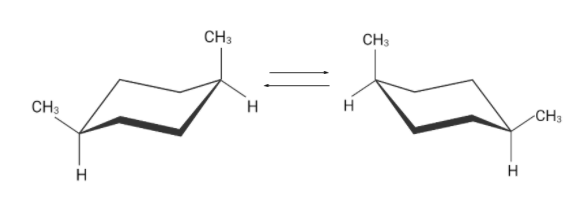
Similarly there are two flipped cyclohexane chairs. These two conformations are identical.
Note :
This compound can be used in various real life applications. The absorption of ultrasonic waves that can be calculated using a reverberation method has been studied using cis-1,4-dimethylcyclohexane.
Complete Step By Step Answer:
Cis-1,4-dimethyl cyclohexane has one chair conformation and two boat conformations. The chair conformation of cyclohexane is more stable than the boat conformation since the $C - H$ bonds are similarly axial and equatorial in the chair conformation, i.e., six of the twelve $C - H$bonds are axial and six are equatorial, and each carbon has one axial and one equatorial $C - H$bond.
The chain structure of cis-1,4-dimethyl cyclohexane

There are two flipped boat conformations . Although both are different but the right one is more stable.

Similarly there are two flipped cyclohexane chairs. These two conformations are identical.

Note :
This compound can be used in various real life applications. The absorption of ultrasonic waves that can be calculated using a reverberation method has been studied using cis-1,4-dimethylcyclohexane.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




