What are the products formed when phenol and nitrobenzene are treated separately with a mixture of concentrated sulfuric acid and concentrated nitric acid?
Answer
639.3k+ views
Hint: To answer this question you must know how benzene and nitrobenzene differ in reaction with a mixture of concentrated sulfuric acid and concentrated nitric acid. In these reactions, ring activation and deactivation plays an important role. Now you have to think about the formation of products.
Complete step by step answer:
When the mixture of concentrated sulphuric acid and concentrated nitric acid reacts together generates the nitronium ion, which acts as an electrophile in electrophilic aromatic substitution reaction.
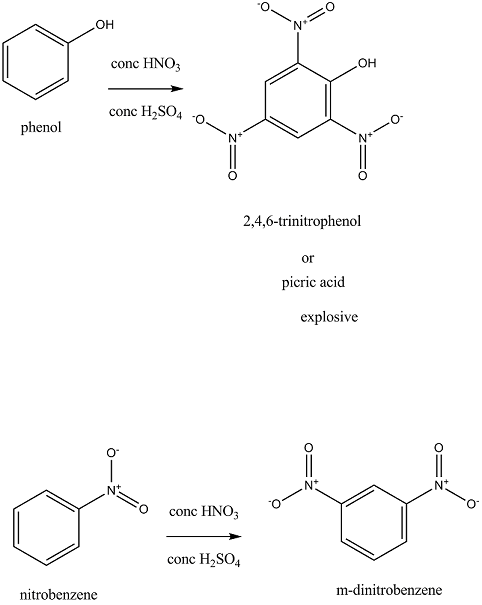
When phenol is treated separately with a mixture of concentrated sulphuric acid and concentrated nitric acid, 2,4,6-trinitrophenol ( also known as picric acid) is obtained. Hydroxyl group is an activating group here and so it is ortho, para directing. In this reaction conditions are so favorable that the nitro group is attached to all ortho and para positions.
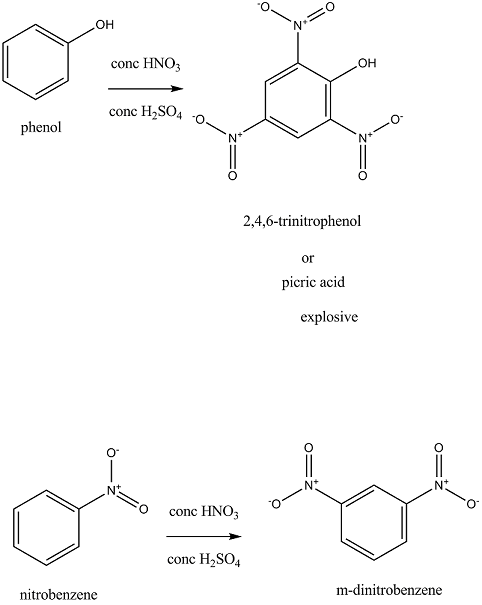
When nitrobenzene is treated separately with a mixture of concentrated sulphuric acid and concentrated nitric acid, m-dinitrobenzene acid is obtained. Nitro group is a deactivating group present here and so it is meta directing.
Therefore, we can conclude that picric acid and m-dinitrobenzene are the products formed when phenol and nitrobenzene are treated separately with a mixture of concentrated sulfuric acid and concentrated nitric acid.
Note: You should also know that the largest use of Picric acid has been in munitions and explosives. Explosive D, also known as Dunnite, is the ammonium salt of picric acid—more powerful but less stable than the more common explosive TNT.
Complete step by step answer:
When the mixture of concentrated sulphuric acid and concentrated nitric acid reacts together generates the nitronium ion, which acts as an electrophile in electrophilic aromatic substitution reaction.
When phenol is treated separately with a mixture of concentrated sulphuric acid and concentrated nitric acid, 2,4,6-trinitrophenol ( also known as picric acid) is obtained. Hydroxyl group is an activating group here and so it is ortho, para directing. In this reaction conditions are so favorable that the nitro group is attached to all ortho and para positions.

When nitrobenzene is treated separately with a mixture of concentrated sulphuric acid and concentrated nitric acid, m-dinitrobenzene acid is obtained. Nitro group is a deactivating group present here and so it is meta directing.

Therefore, we can conclude that picric acid and m-dinitrobenzene are the products formed when phenol and nitrobenzene are treated separately with a mixture of concentrated sulfuric acid and concentrated nitric acid.
Note: You should also know that the largest use of Picric acid has been in munitions and explosives. Explosive D, also known as Dunnite, is the ammonium salt of picric acid—more powerful but less stable than the more common explosive TNT.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




