What are the definitions of condensation reaction?
Answer
529.2k+ views
Hint: A condensation reaction in organic chemistry is the joining of two molecules to produce a single molecule, usually with the loss of a minor molecule like water. The reaction is also known as a dehydration synthesis when water is lost. Other molecules, such as ammonia, ethanol, acetic acid, and hydrogen sulphide, can also be lost.
Complete answer:
The two molecules are normally added to the addition product in a step-by-step method, usually in equilibrium and with the loss of a water molecule (hence the name condensation). A diverse family of reactions that can occur in acidic or basic circumstances or in the presence of a catalyst, the reaction may otherwise include the functional groups of the molecule. This group of events is necessary for the creation of peptide bonds between amino acids and the production of fatty acids, therefore thus is a necessary part of life.
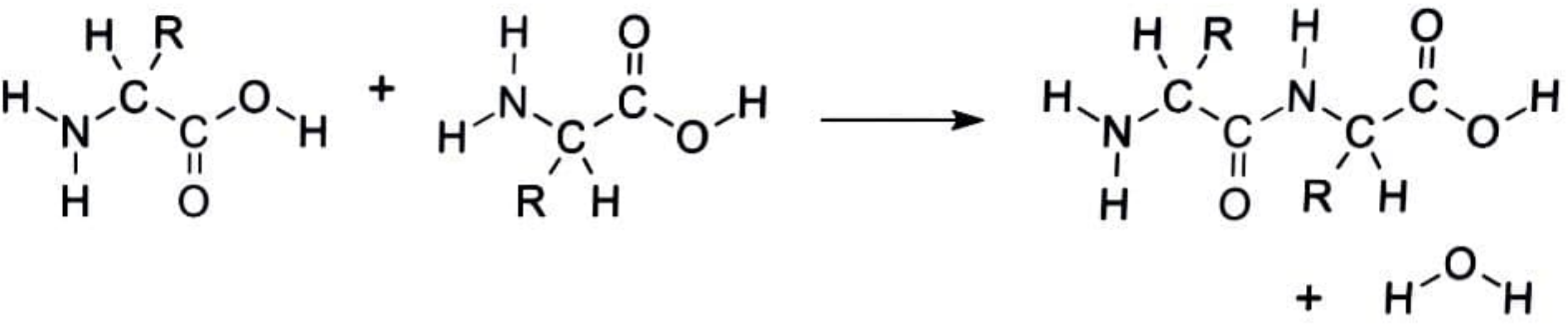
fig.condensation of two amino acids to give a peptide bond.
Condensation reactions can be in a variety of forms. Aldol condensation and Knoevenagel condensation, both of which produce water as a by-product, as well as the Claisen condensation and the Dieckman condensation (intramolecular Claisen condensation), all of which produce alcohols as by-products, are common examples.
Note:
A condensation reaction in organic chemistry is the joining of two molecules to produce a single molecule, usually with the loss of a minor molecule like water. The reaction is also known as a dehydration synthesis when water is lost.
Complete answer:
The two molecules are normally added to the addition product in a step-by-step method, usually in equilibrium and with the loss of a water molecule (hence the name condensation). A diverse family of reactions that can occur in acidic or basic circumstances or in the presence of a catalyst, the reaction may otherwise include the functional groups of the molecule. This group of events is necessary for the creation of peptide bonds between amino acids and the production of fatty acids, therefore thus is a necessary part of life.

fig.condensation of two amino acids to give a peptide bond.
Condensation reactions can be in a variety of forms. Aldol condensation and Knoevenagel condensation, both of which produce water as a by-product, as well as the Claisen condensation and the Dieckman condensation (intramolecular Claisen condensation), all of which produce alcohols as by-products, are common examples.
Note:
A condensation reaction in organic chemistry is the joining of two molecules to produce a single molecule, usually with the loss of a minor molecule like water. The reaction is also known as a dehydration synthesis when water is lost.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




