What are microstates in chemistry?
Answer
533.7k+ views
Hint: The microstate is the arrangement or specific way of energy of each molecule in the system at one kind of instance. It is something like a theoretical instantaneous photo of the location of a molecule and atom in the macro-state.
Complete answer:
Let us understand the concept in detail-
Microstates, basically, are the microscopic ways to observe certain processes taking place in the system. These microstates describe the location and arrangement of micro molecules within the macro-state. The visual location is captured and thus is described as a microstate. An example is shown below-
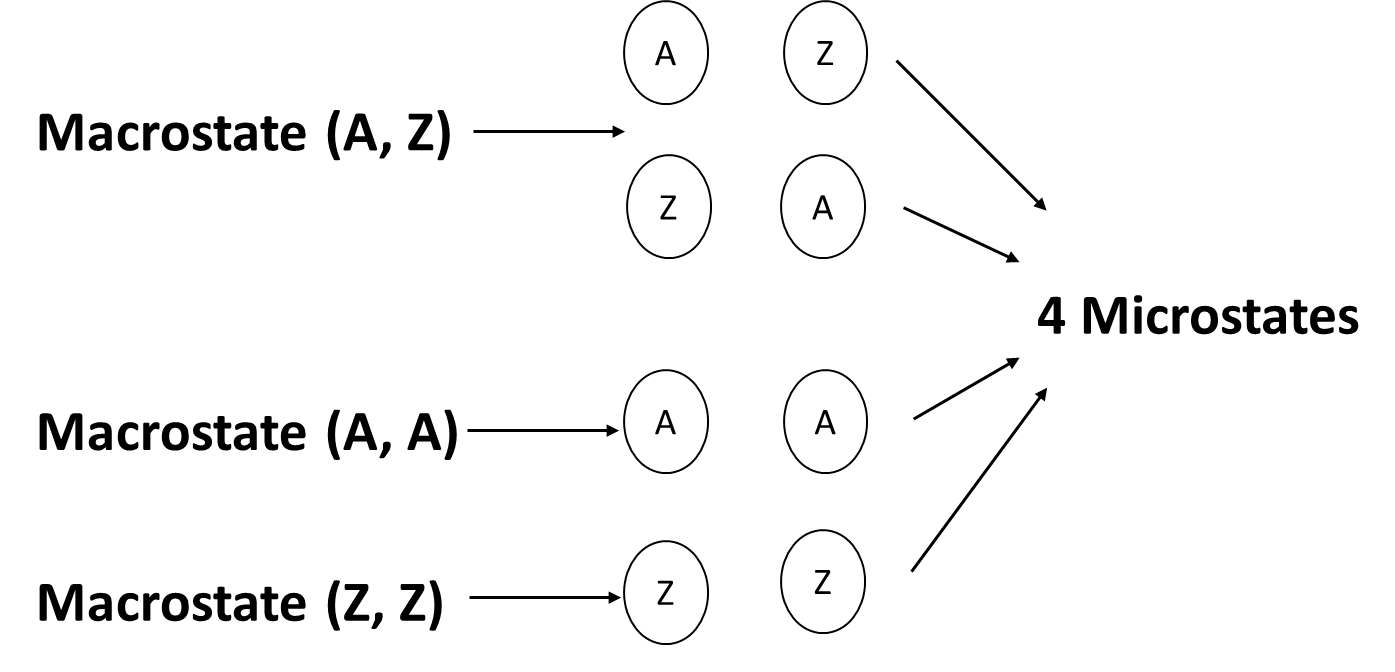
Analogous to this, there is a macro-state which is the matter which is large enough for us to measure i.e., which is present in bulk. For example, volume, pressure or temperature.
Note:
Factually, macro means something that is large whereas micro means something which is comparatively smaller. In chemical thermodynamics, these terms do not have the relatively stated English meaning. Instead, they are the two different ways of looking towards the working system.
The microstate is one of the arrangements of molecules for a particular macro-state.
Complete answer:
Let us understand the concept in detail-
Microstates, basically, are the microscopic ways to observe certain processes taking place in the system. These microstates describe the location and arrangement of micro molecules within the macro-state. The visual location is captured and thus is described as a microstate. An example is shown below-

Analogous to this, there is a macro-state which is the matter which is large enough for us to measure i.e., which is present in bulk. For example, volume, pressure or temperature.
Note:
Factually, macro means something that is large whereas micro means something which is comparatively smaller. In chemical thermodynamics, these terms do not have the relatively stated English meaning. Instead, they are the two different ways of looking towards the working system.
The microstate is one of the arrangements of molecules for a particular macro-state.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




