What are Claisen Schmidt's condensation and Kolbe's Reaction? Give one example of each.
Answer
623.7k+ views
Hint: Claisen Schmidt condensation is the chemical property of aldehyde and ketone. It is based on a cross aldol condensation reaction. Kolbe's reaction is based on the chemical property of phenol. Salicylic acid is the major product of Kolbe's reaction.
Complete answer:
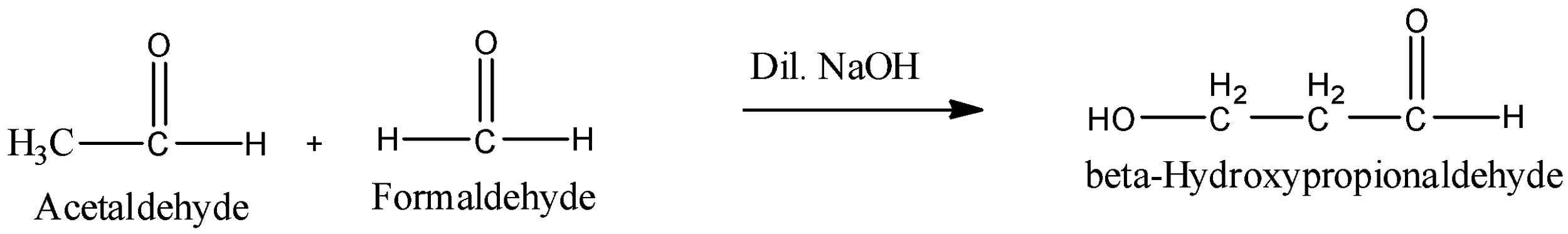
Aldol condensation is not confined to the condensation of two molecules of the same aldehyde or ketone (self-condensation). It can also take place between two different aldehydes or ketones or between one aldehyde and one ketone. Such an aldol condensation between two different aldehydes or two ketones or between one aldehyde and one ketone is called the cross aldol condensation. For example Reaction between Acetaldehyde and Formaldehyde forms beta- hydroxypropionaldehyde.
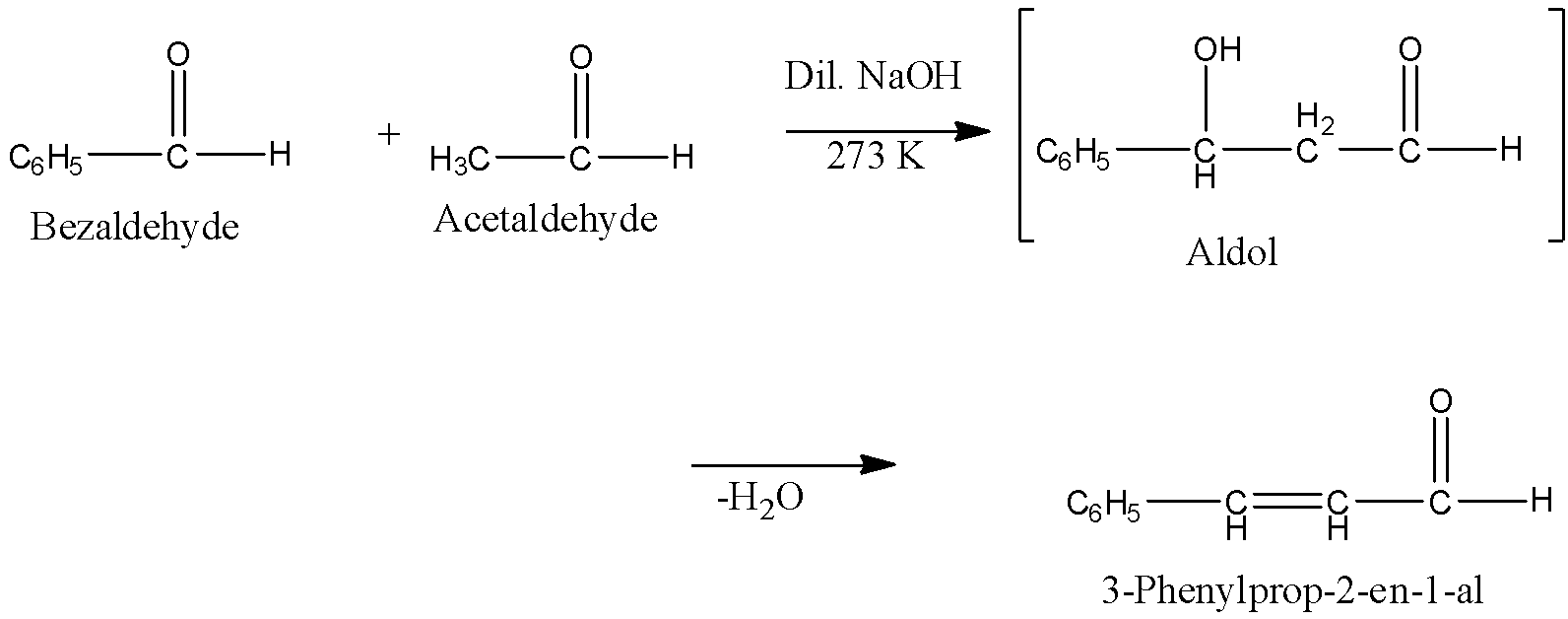
Claisen Schmidt condensation: When a base-catalyzed cross aldol condensation between an aromatic aldehyde and an aliphatic aldehyde or a ketone is called Claisen Schmidt condensation or simply Claisen reaction. For example, Benzaldehyde reacts with Acetaldehyde to form 3-Phenylprop-2-en-1-al.
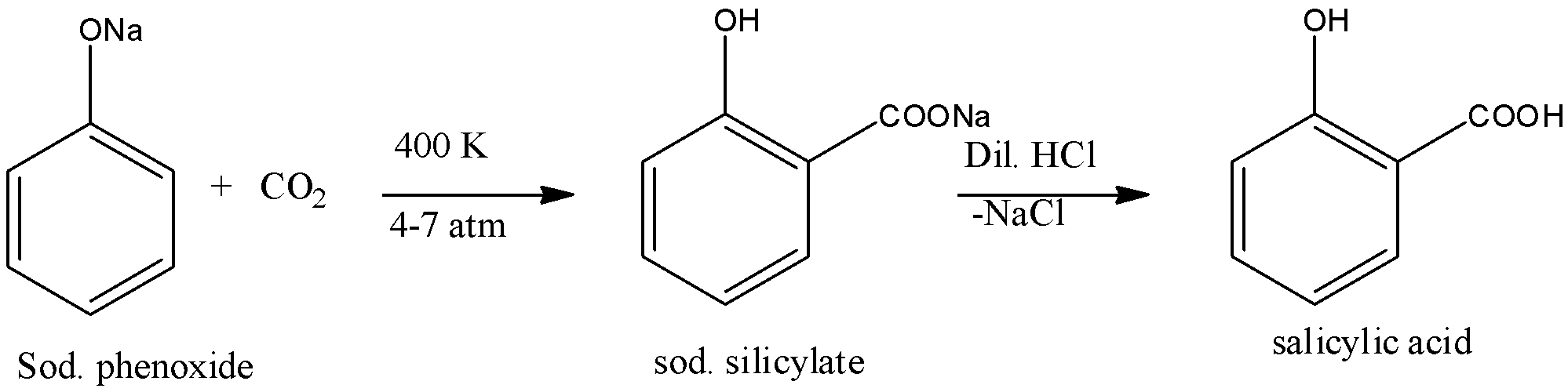
Kolbe’s Reaction: Sodium phenoxide when heated with carbon dioxide at 400 K under a pressure of 4-7 atmospheres followed by acidification gives 2-hydroxybenzoic acid ( salicylic acid) as the main product with a small amount of 4-hydroxybenzoic acid. This reaction is called Kolbe’s reaction. The reaction is given below:
Note: The salicylic acid which is prepared from Kolbe's reaction is used for the preparation of aspirin, which is widely used for relieving pain and to bring down the body temperature. Even aliphatic esters containing alpha-hydrogen atoms can undergo Claisen Schmidt Condensation on treatment with an aromatic aldehyde in the presence of a base.
Complete answer:
Aldol condensation is not confined to the condensation of two molecules of the same aldehyde or ketone (self-condensation). It can also take place between two different aldehydes or ketones or between one aldehyde and one ketone. Such an aldol condensation between two different aldehydes or two ketones or between one aldehyde and one ketone is called the cross aldol condensation. For example Reaction between Acetaldehyde and Formaldehyde forms beta- hydroxypropionaldehyde.

Claisen Schmidt condensation: When a base-catalyzed cross aldol condensation between an aromatic aldehyde and an aliphatic aldehyde or a ketone is called Claisen Schmidt condensation or simply Claisen reaction. For example, Benzaldehyde reacts with Acetaldehyde to form 3-Phenylprop-2-en-1-al.

Kolbe’s Reaction: Sodium phenoxide when heated with carbon dioxide at 400 K under a pressure of 4-7 atmospheres followed by acidification gives 2-hydroxybenzoic acid ( salicylic acid) as the main product with a small amount of 4-hydroxybenzoic acid. This reaction is called Kolbe’s reaction. The reaction is given below:

Note: The salicylic acid which is prepared from Kolbe's reaction is used for the preparation of aspirin, which is widely used for relieving pain and to bring down the body temperature. Even aliphatic esters containing alpha-hydrogen atoms can undergo Claisen Schmidt Condensation on treatment with an aromatic aldehyde in the presence of a base.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




