Why are all P-F bonds in $P{F_5}$ are not equivalent?
A.$P{F_5}$ has $s{p^3}d$ hybridisation, out of five $P - F$ bonds three are equatorial which have different lengths.
B.$P{F_5}$ has $s{p^3}$ hybridisation, out of $P - F$ bonds two are equatorial which have different lengths.
C.Out of five $P - F$ bonds two are axial and three equatorial. All five bonds have different bond lengths.
D.$P{F_5}$ is made up of two types of bond namely covalent and coordinate, hence are not equivalent.
Answer
542.2k+ views
Hint: VSEPR Theory (valence shell electron pair repulsion theory) plays an important role in predicting shape of a molecule. $P{F_5}$ has $5$ bond pairs of electrons and $0$lone pairs of electrons which will be needed to know its shape. It’s hybridization is $s{p^3}d$ .
Complete step by step solution:
Bonds to a non ring atom with an angle of about ${90^ \circ }$ to the ring plane are termed axial.
Bonds to a non ring atom which make only a small angle compared with the plane of the ring are termed as equatorial bonds.
$P{F_5}$ is phosphorus pentafluoride. It has a $5$ bond pair and a $0$ lone pair. VSEPR Theory is used to predict the geometry of individual molecules from the number of electron pairs surrounding the central atom. According to this theory if a compound has $5$ bond pair and $0$ lone pair. Then it will have trigonal bipyramidal geometry.
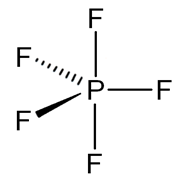
This kind of geometry has $s{p^3}d$ hybridization. This means $P{F_5}$ has trigonal bipyramidal geometry in which 3 bonds are at equatorial position and two bonds are at axial position. Bonds at axial position are larger in length because they experience more repulsion by 3 equatorial $P - F$ bonds, that is why all $P - F$ bonds in $P{F_5}$ are not equivalent.
So our answer to this question is option C that is Out of five $P - F$ bonds two are axial and three equatorial. All five bonds have different bond lengths.
Additional information: To explain the concept of shapes of molecules clearly hybridization was introduced. It involves intermixing of two or more atomic orbitals of slightly different energies but of the same atom so that a redistribution of energy takes place between them resulting in the formation of an equal number of new orbitals which are called hybrid orbitals which will have the same energy, size and shape.
Note: $P{F_5}$ is a neutral and non polar compound (compound having symmetric distribution of charge so that net charge is zero on compound) with $5$ fluorine atoms attached to central phosphorus element and all these fluorine atoms are attached symmetrically around phosphorus metal.
Complete step by step solution:
Bonds to a non ring atom with an angle of about ${90^ \circ }$ to the ring plane are termed axial.
Bonds to a non ring atom which make only a small angle compared with the plane of the ring are termed as equatorial bonds.
$P{F_5}$ is phosphorus pentafluoride. It has a $5$ bond pair and a $0$ lone pair. VSEPR Theory is used to predict the geometry of individual molecules from the number of electron pairs surrounding the central atom. According to this theory if a compound has $5$ bond pair and $0$ lone pair. Then it will have trigonal bipyramidal geometry.

This kind of geometry has $s{p^3}d$ hybridization. This means $P{F_5}$ has trigonal bipyramidal geometry in which 3 bonds are at equatorial position and two bonds are at axial position. Bonds at axial position are larger in length because they experience more repulsion by 3 equatorial $P - F$ bonds, that is why all $P - F$ bonds in $P{F_5}$ are not equivalent.
So our answer to this question is option C that is Out of five $P - F$ bonds two are axial and three equatorial. All five bonds have different bond lengths.
Additional information: To explain the concept of shapes of molecules clearly hybridization was introduced. It involves intermixing of two or more atomic orbitals of slightly different energies but of the same atom so that a redistribution of energy takes place between them resulting in the formation of an equal number of new orbitals which are called hybrid orbitals which will have the same energy, size and shape.
Note: $P{F_5}$ is a neutral and non polar compound (compound having symmetric distribution of charge so that net charge is zero on compound) with $5$ fluorine atoms attached to central phosphorus element and all these fluorine atoms are attached symmetrically around phosphorus metal.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

Which among the following are examples of coming together class 11 social science CBSE

Discuss the various forms of bacteria class 11 biology CBSE

What organs are located on the left side of your body class 11 biology CBSE

Draw a labelled diagram showing structure of LS of class 11 biology CBSE




