When any substance (except water) is heated, its density:
$
{\text{A}}{\text{. increases}} \\
{\text{B}}{\text{. decreases}} \\
{\text{C}}{\text{. remains same}} \\
{\text{D}}{\text{. none of these}} \\
$
Answer
628.5k+ views
Hint: Density is the ratio of mass and volume of the substance. So the change caused by temperature on mass and volume should be checked, to now the effect on density.
Complete step-by-step answer:
Mass is a property that is fixed. It does not change unless relativity applies. So, on increasing temperature, the mass remains unaffected.
In general, all substances (except water) expand on heating. As on heating, the internal energy of a substance increases. There are vibrations in molecules and therefore, the molecular distances increase. As the distance between molecules is increasing, the substance occupies more space. Hence, as the temperature is increased so their volume increases.
So, we can generalize that:
With an increase in temperature:
Mass remains unaffected
Volume increases
As, $density = \dfrac{{mass}}{{volume}}$
In the diffraction, the numerator remains constant but the denominator is increasing. As a whole, the diffraction (=density) is decreasing.
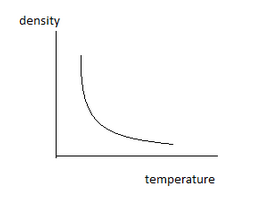
So density decreases.
Correct option is (B).
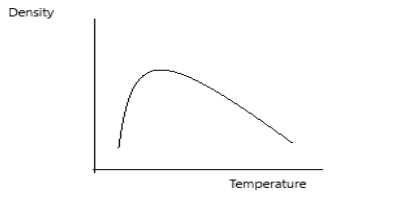
Note: In case of water, the expansion of volume follows a different trend. So the variation in density with temperature will be different.
The variation in density for water can be shown as:
Complete step-by-step answer:
Mass is a property that is fixed. It does not change unless relativity applies. So, on increasing temperature, the mass remains unaffected.
In general, all substances (except water) expand on heating. As on heating, the internal energy of a substance increases. There are vibrations in molecules and therefore, the molecular distances increase. As the distance between molecules is increasing, the substance occupies more space. Hence, as the temperature is increased so their volume increases.
So, we can generalize that:
With an increase in temperature:
Mass remains unaffected
Volume increases
As, $density = \dfrac{{mass}}{{volume}}$
In the diffraction, the numerator remains constant but the denominator is increasing. As a whole, the diffraction (=density) is decreasing.

So density decreases.
Correct option is (B).
Note: In case of water, the expansion of volume follows a different trend. So the variation in density with temperature will be different.
The variation in density for water can be shown as:

Recently Updated Pages
Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Computer Science: Engaging Questions & Answers for Success

Class 10 Question and Answer - Your Ultimate Solutions Guide

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Who Won 36 Oscar Awards? Record Holder Revealed

What is the median of the first 10 natural numbers class 10 maths CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

Make a sketch of the human nerve cell What function class 10 biology CBSE

Define Potential, Developed, Stock and Reserved resources




