An element A is tetravalent and another element B is divalent. The formula of the compound formed by the combination of these elements is:
A. \[{A_2}B\]
B. \[AB\]
C. \[A{B_2}\]
D. \[{A_2}{B_3}\]
Answer
615k+ views
Hint: The molecular formula of a compound represents the atoms and the number of atoms present in the compound. The number of the atoms in the compound are written in the form of suffix. A structural formula of a compound represents the structure of a particular compound.
Complete step by step answer:
We are given that an element A is tetravalent, hence its oxidation number can be \[{{\text{A}}^{{\text{ + 4}}}}\]
Also, it is also given that element B is divalent hence its oxidation number can be \[{{\text{B}}^{{\text{2}} - }}\].
Therefore, we give the formula of the compound by cross method as,
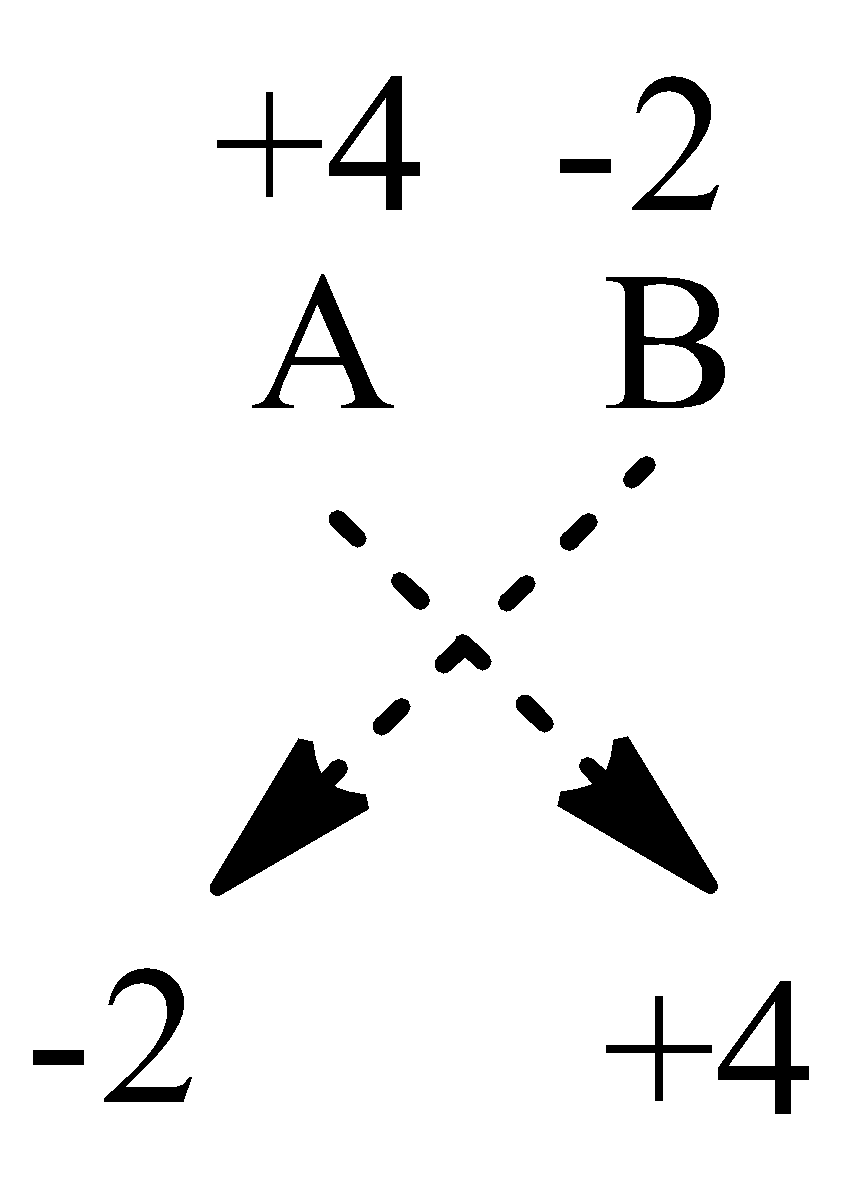
So, the formula of the compound will be \[{{\text{A}}_{\text{2}}}{{\text{B}}_{\text{4}}}\]or \[{\text{A}}{{\text{B}}_2}\].
So, the correct answer is Option C .
Note:
In chemical compounds, the elements are present in a fixed ratio by mass. This was given by Joseph Proust. This is the “law of constant proportions''. This “law of constant proportions” is also termed as the law of definite proportions or as the Proust’s law. For example, pure water contains oxygen and hydrogen in a fixed ratio of 1:8. Water contains 0.88 grams of oxygen and 0.11 grams of hydrogen
Students may get confused between formula units and molecular formulas. By formula unit we represent the ratio of the ions which is present in a compound. The molecular formula of a compound represents the atoms and the number of atoms present in the compound. The number of the atoms in the compound are written in the form of suffix.
Complete step by step answer:
We are given that an element A is tetravalent, hence its oxidation number can be \[{{\text{A}}^{{\text{ + 4}}}}\]
Also, it is also given that element B is divalent hence its oxidation number can be \[{{\text{B}}^{{\text{2}} - }}\].
Therefore, we give the formula of the compound by cross method as,

So, the formula of the compound will be \[{{\text{A}}_{\text{2}}}{{\text{B}}_{\text{4}}}\]or \[{\text{A}}{{\text{B}}_2}\].
So, the correct answer is Option C .
Note:
In chemical compounds, the elements are present in a fixed ratio by mass. This was given by Joseph Proust. This is the “law of constant proportions''. This “law of constant proportions” is also termed as the law of definite proportions or as the Proust’s law. For example, pure water contains oxygen and hydrogen in a fixed ratio of 1:8. Water contains 0.88 grams of oxygen and 0.11 grams of hydrogen
Students may get confused between formula units and molecular formulas. By formula unit we represent the ratio of the ions which is present in a compound. The molecular formula of a compound represents the atoms and the number of atoms present in the compound. The number of the atoms in the compound are written in the form of suffix.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




