An atom of an element has $\,4\,$ electrons in the outermost $\,M\,$ shell. What will be the atomic number of this element? Name this element. Find the valency of this element. Draw a schematic diagram of this atom showing the distribution of electrons in its shells.
Answer
610.2k+ views
Hint: Atomic number is numerically equal to the number of electrons in a neutral atom. If we can find the atomic number, we can search this element in the periodic table and can find its name and simultaneously the valency can also be found out.
Complete step by step answer:
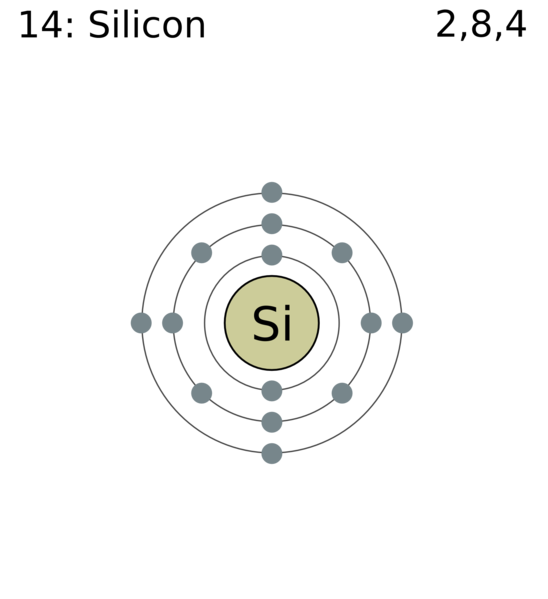
Let’s first find out the atomic number of this element;
In this element it is said that the outermost shell is the $\,M\,$ shell. So, this is the third shell in an atom. $\,K\,$ and $\,L\,$ are the first two shells respectively and the maximum number of electrons that can be accommodated in these two shells are $\,2\,$ and $\,8\,$ respectively. So, the atomic number is equal to;
$\,2 + 8 + 4 = 14\,$. When we search this element in the periodic-table we can see that this element is silicon.
Now, valency is equal to the number of electrons in the outermost shell and hence its valency is equal to $\,4\,$.
Additional information: The word silicon comes from the Latin silex or silicis, meaning "flint" or "hard stone." On a weight basis, only oxygen is surpassed by the concentration of silicon in the crust of Earth. Silicon is part of the carbon family and is also a non-metallic element.
Note:
Note that the maximum accommodation capacity for each shell is $\,8\,$ except the first shell which is the $\,K\,$ shell. If an atom's outermost shell contains a limit of 8 electrons, so a complete octet is assumed to have been reached by the atom. Valence electrons are active in any chemical reaction and they typically produce more energy in the outermost orbit than the electrons found in other orbits.
Complete step by step answer:
Let’s first find out the atomic number of this element;
In this element it is said that the outermost shell is the $\,M\,$ shell. So, this is the third shell in an atom. $\,K\,$ and $\,L\,$ are the first two shells respectively and the maximum number of electrons that can be accommodated in these two shells are $\,2\,$ and $\,8\,$ respectively. So, the atomic number is equal to;
$\,2 + 8 + 4 = 14\,$. When we search this element in the periodic-table we can see that this element is silicon.
Now, valency is equal to the number of electrons in the outermost shell and hence its valency is equal to $\,4\,$.

Additional information: The word silicon comes from the Latin silex or silicis, meaning "flint" or "hard stone." On a weight basis, only oxygen is surpassed by the concentration of silicon in the crust of Earth. Silicon is part of the carbon family and is also a non-metallic element.
Note:
Note that the maximum accommodation capacity for each shell is $\,8\,$ except the first shell which is the $\,K\,$ shell. If an atom's outermost shell contains a limit of 8 electrons, so a complete octet is assumed to have been reached by the atom. Valence electrons are active in any chemical reaction and they typically produce more energy in the outermost orbit than the electrons found in other orbits.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




