Among the three isomers of the nitrophenol, the one that is least soluble in water is:
(a) m-nitrophenol
(b) o-nitrophenol
(c) p-nitrophenol
(d) none of these
Answer
630.9k+ views
Hint: Some of the organic compounds can form effective hydrogen bonds. Due to such hydrogen bonds, it can decrease the solubility of that compound.
Complete step by step solution:
Organic compounds can form intermolecular hydrogen bonds. Intra hydrogen bonding is hydrogen bond formation within the molecule. It generally leads to cyclisation of the molecule and prevents its association with other molecules as well prevents it from dissociating in water. It renders its activity.
Let us now consider each option.
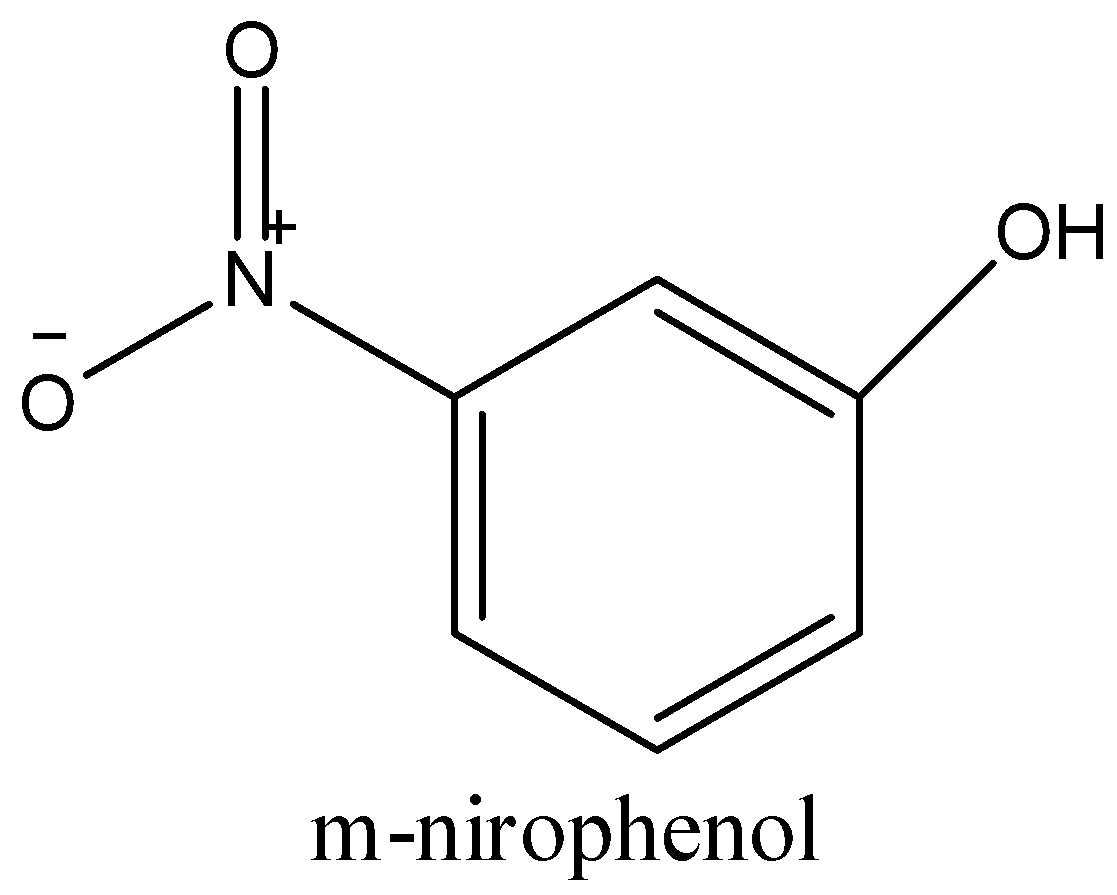
-m-nitrophenol has hydroxyl group in the meta position of the benzene ring of nitrophenol. There is distance between the OH- group and the nitro group and cannot form any intermolecular hydrogen bond. So, it is soluble in water due to the presence of free hydroxyl and nitro groups.
-In o-nitrophenol, the hydroxyl group is in the ortho position of nitrobenzene; that is; it is next to the nitro group. The hydrogen of the hydroxyl group can easily form an intermolecular hydrogen bond with the oxygen of the nitro group as shown in the figure below.
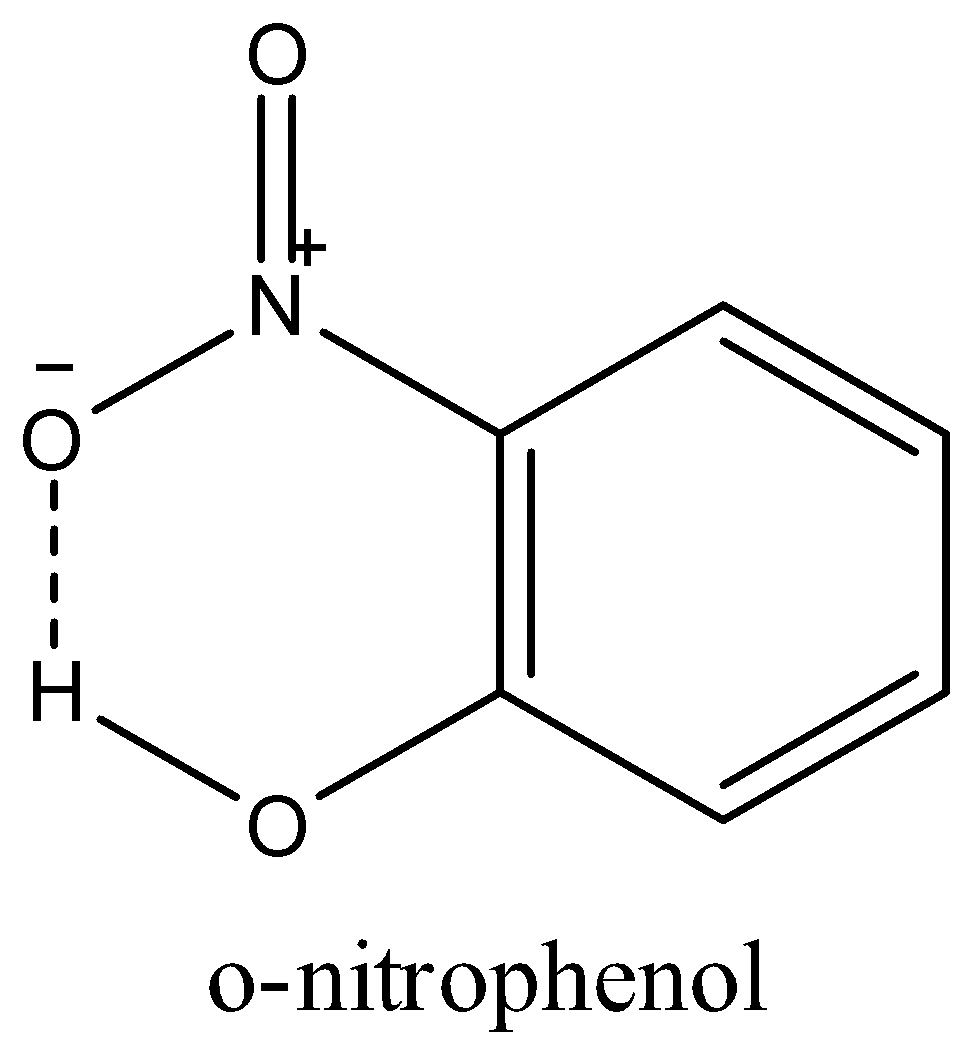
So, the nitro group and the hydroxyl groups are not free and o-nitrophenol will not get soluble in water.
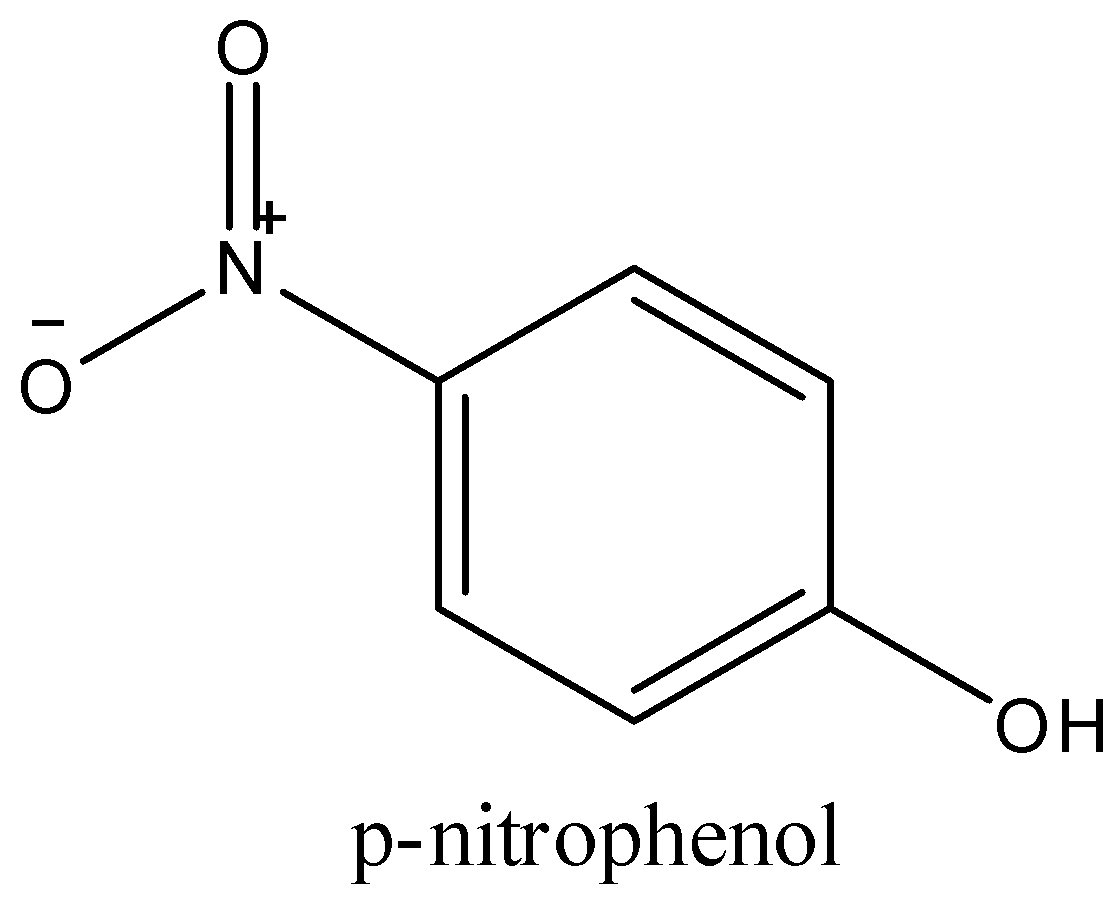
-p-nitrophenol, in this the hydroxyl group is on the para position and the nitro group and hydroxyl groups cannot form any bonds between them. They are free, and p-nitrophenol is soluble in water.
Thus, o-nitrophenol is least soluble in water and the correct option is (b).
Note: In p-nitrophenol intermolecular hydrogen bonding occurs not intramolecular. Intermolecular hydrogen bonding is the hydrogen bond between two different molecules.
Complete step by step solution:
Organic compounds can form intermolecular hydrogen bonds. Intra hydrogen bonding is hydrogen bond formation within the molecule. It generally leads to cyclisation of the molecule and prevents its association with other molecules as well prevents it from dissociating in water. It renders its activity.
Let us now consider each option.
-m-nitrophenol has hydroxyl group in the meta position of the benzene ring of nitrophenol. There is distance between the OH- group and the nitro group and cannot form any intermolecular hydrogen bond. So, it is soluble in water due to the presence of free hydroxyl and nitro groups.

-In o-nitrophenol, the hydroxyl group is in the ortho position of nitrobenzene; that is; it is next to the nitro group. The hydrogen of the hydroxyl group can easily form an intermolecular hydrogen bond with the oxygen of the nitro group as shown in the figure below.

So, the nitro group and the hydroxyl groups are not free and o-nitrophenol will not get soluble in water.
-p-nitrophenol, in this the hydroxyl group is on the para position and the nitro group and hydroxyl groups cannot form any bonds between them. They are free, and p-nitrophenol is soluble in water.

Thus, o-nitrophenol is least soluble in water and the correct option is (b).
Note: In p-nitrophenol intermolecular hydrogen bonding occurs not intramolecular. Intermolecular hydrogen bonding is the hydrogen bond between two different molecules.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




