Aliphatic cyclic compounds are known as:
(A) Alicyclic compounds
(B) Polycyclic compounds
(C) Aromatic compounds
(D) Heterocyclic compounds
Answer
611.7k+ views
Hint: Cyclic compounds are the compounds that have a ring like structure. Aliphatic compounds are the compounds that do not have aromatic character. Cycloalkanes are the examples of aliphatic cyclic compounds.
Complete step by step solution:
Let’s know about aliphatic cyclic compounds.
- Cyclic compounds are the compounds that include a cyclic structure. Thus, one or more rings may be there in cyclic compounds. Aliphatic compounds are the compounds that do not have any aromatic character.
- Thus, aliphatic cyclic compounds have a cyclic structure as well as absence of conjugated double bonds. So, they are also called alicyclic compounds.
e.g. Cyclohexane
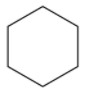
- Polycyclic compounds are the aliphatic cyclic compounds which always have more than one ring in their structure. They do not have aromatic rings in their structure.
- Aromatic compounds have conjugated double bonds in their structure. All the aromatic compounds have ring structure. They may have one or more rings in their structure. They always have $4n + 2{\text{ }}\pi $ electrons in their structure where n is the number of rings present in the structure.
- Heterocyclic compounds are the cyclic compounds which involve heteroatoms in the ring structure. The number of heteroatoms present in the ring may be one or more. They may be aromatic or not. Heteroatoms include atoms like N, O, P, S, etc...
- Thus, we can conclude that aliphatic cyclic compounds are known as alicyclic compounds.
So, the correct answer is (A).
Note: Remember that the ring or cycle in the alicyclic compounds do not have any heteroatoms like S, P, N, or O. All the atoms forming the cyclic structure are carbons. The other atoms may be present in the side chain but not in the ring forming skeleton.
Complete step by step solution:
Let’s know about aliphatic cyclic compounds.
- Cyclic compounds are the compounds that include a cyclic structure. Thus, one or more rings may be there in cyclic compounds. Aliphatic compounds are the compounds that do not have any aromatic character.
- Thus, aliphatic cyclic compounds have a cyclic structure as well as absence of conjugated double bonds. So, they are also called alicyclic compounds.
e.g. Cyclohexane

- Polycyclic compounds are the aliphatic cyclic compounds which always have more than one ring in their structure. They do not have aromatic rings in their structure.
- Aromatic compounds have conjugated double bonds in their structure. All the aromatic compounds have ring structure. They may have one or more rings in their structure. They always have $4n + 2{\text{ }}\pi $ electrons in their structure where n is the number of rings present in the structure.
- Heterocyclic compounds are the cyclic compounds which involve heteroatoms in the ring structure. The number of heteroatoms present in the ring may be one or more. They may be aromatic or not. Heteroatoms include atoms like N, O, P, S, etc...
- Thus, we can conclude that aliphatic cyclic compounds are known as alicyclic compounds.
So, the correct answer is (A).
Note: Remember that the ring or cycle in the alicyclic compounds do not have any heteroatoms like S, P, N, or O. All the atoms forming the cyclic structure are carbons. The other atoms may be present in the side chain but not in the ring forming skeleton.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




