(a) What is soap?
(b) Describe the structure of a soap molecule with the help of a diagram.
(c) Explain the cleansing action of soap. Draw a diagram to illustrate your answer.
Answer
601.8k+ views
Hint: We know that soaps are very important in life. We usually wash our hands using soap to remove dirt. It helps in cleansing our body, clothes etc. It has a cleansing property. It saves us from bacteria which are harmful.
Complete step by step solution:
(a) The sodium and potassium salts of the long chain carboxylic acids are referred to as soaps. It consists of a long hydrocarbon chain having carboxylic acid on one end. This has an ionic bond with metal ions which is usually sodium or potassium.
(b) The structure of a soap molecule is given below:
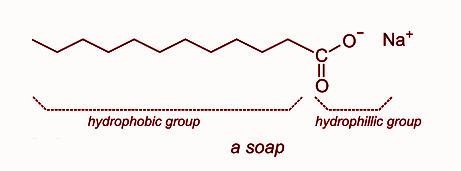
This is the structure of sodium stearate which is an example of soap molecule. It has a hydrophilic and hydrophobic part in it. The hydrophobic end is nonpolar and insoluble in water. In the above molecule, the hydrophobic part is ${\text{ - }}{{\text{C}}_{17}}{{\text{H}}_{35}}$. The hydrophilic end is polar and soluble in water. In the molecule, the hydrophilic end is $ - {\text{COONa}}$.
(c) Micelles is the name of soap molecules. Hydrophobic end is attached to the dust, oil, grease, dirt or the materials which we want to remove, detaching takes place, gets suspended in water and can easily be removed.
When a dirt is put in water containing soap, then the non-polar end of soap in micelle attach to an oil particle present on the surface of dirt. The soap micelles trap the oily particles by the nonpolar end. The polar end attaches to the water when the dirt is agitated in soap solution.
The structure of a micelle and the cleansing action of soap molecule is given below:
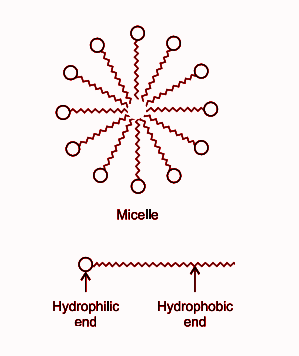
Note: There is a chance of making a mistake that we may confuse soaps with detergent. The main difference between soap and detergent is that the cleansing action of soap does not work properly with hard water. While detergent works well with hard water too.
Complete step by step solution:
(a) The sodium and potassium salts of the long chain carboxylic acids are referred to as soaps. It consists of a long hydrocarbon chain having carboxylic acid on one end. This has an ionic bond with metal ions which is usually sodium or potassium.
(b) The structure of a soap molecule is given below:

This is the structure of sodium stearate which is an example of soap molecule. It has a hydrophilic and hydrophobic part in it. The hydrophobic end is nonpolar and insoluble in water. In the above molecule, the hydrophobic part is ${\text{ - }}{{\text{C}}_{17}}{{\text{H}}_{35}}$. The hydrophilic end is polar and soluble in water. In the molecule, the hydrophilic end is $ - {\text{COONa}}$.
(c) Micelles is the name of soap molecules. Hydrophobic end is attached to the dust, oil, grease, dirt or the materials which we want to remove, detaching takes place, gets suspended in water and can easily be removed.
When a dirt is put in water containing soap, then the non-polar end of soap in micelle attach to an oil particle present on the surface of dirt. The soap micelles trap the oily particles by the nonpolar end. The polar end attaches to the water when the dirt is agitated in soap solution.
The structure of a micelle and the cleansing action of soap molecule is given below:

Note: There is a chance of making a mistake that we may confuse soaps with detergent. The main difference between soap and detergent is that the cleansing action of soap does not work properly with hard water. While detergent works well with hard water too.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Find the value of the expression given below sin 30circ class 11 maths CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

What organs are located on the left side of your body class 11 biology CBSE

Difference between physical and chemical change class 11 chemistry CBSE

Which of the following is the hottest month AMay BJune class 11 social science CBSE

What is the percentage of photosynthetically active class 11 biology CBSE




