A uniform cylinder of length L and M having cross-sectional area A is suspended, with its length vertical, from a fixed point by a massless spring, such that it is half submerged in liquid of density $\sigma$ at equilibrium position. The extension ${{x}_{0}}$ of the spring when it is in equilibrium, is
A. $\dfrac{Mg}{k}\left( 1-\dfrac{LA\sigma }{M} \right)$
B. $\dfrac{Mg}{k}\left( 1-\dfrac{LA\sigma }{2M} \right)$
C. $\dfrac{Mg}{k}\left( 1+\dfrac{LA\sigma }{M} \right)$
D. $\dfrac{Mg}{k}$
Answer
577.8k+ views
Hint:First, identify the forces acting on the given cylinder. Then find the magnitude and direction of each force and find the net force acting on the cylinder. Since the cylinder is in equilibrium, equate the net force to zero.
Formula used:
${{F}_{g}}=Mg$
${{F}_{B}}={{V}_{s}}g\sigma$
${{F}_{s}}=k{{x}_{0}}$
Complete step by step answer:
At the equilibrium position, the uniform cylinder will be standing straight up.
There will be three forces acting on the cylinder and that are the gravitational force, the spring force and the force of buoyancy exerted by the liquid.
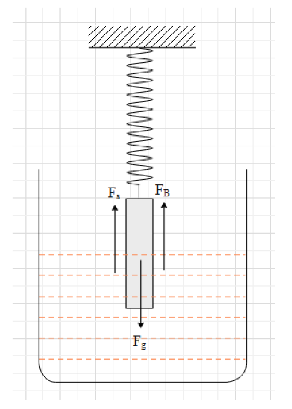
The earth exerts the gravitational force. The gravitational force on a body of mass m is given as ${{F}_{g}}=mg$, where g is the acceleration due to gravity. The gravitational force on the body is always direction downwards.
This means that in the case, the gravitational force on the cylinder is equal to ${{F}_{g}}=Mg$ and is
directed in the downward direction.
When a body is submerged in a liquid, the liquid exerts an upward force on the body called buoyant force. The magnitude of the buoyant force is given as ${{F}_{B}}={{V}_{s}}g\sigma$,
where ${{V}_{s}}$ is the volume of the body submerged in the liquid and $\sigma$ is the density of the liquid.
It is given that half of the cylinder is submerged in the liquid and the volume of a cylinder is equal to $V=AL$, where A is the cross sectional area and L is its length.
This means that ${{F}_{B}}=ALg\sigma$ and is directed upwards.
The cylinder is given to be in equilibrium. Therefore, the cylinder will be at rest and the net force on it will be zero.
Let the extension in the spring be ${{x}_{0}}$. Therefore, the spring force exerted on the cylinder is equal to ${{F}_{s}}=k{{x}_{0}}$, where k is the spring constant of the spring. The spring will be in the upward direction.
Therefore, the net force on the cylinder is ${{F}_{net}}={{F}_{s}}+{{F}_{B}}-{{F}_{g}}$.
$\Rightarrow {{F}_{net}}=k{{x}_{0}}+ALg\sigma -Mg$.
But we know that ${{F}_{net}}=0$.
$\Rightarrow k{{x}_{0}}+ALg\sigma -Mg=0$
$\Rightarrow {{x}_{0}}=\dfrac{Mg-ALg\sigma }{k}$
Take $\dfrac{Mg}{k}$ as a common term.
$\Rightarrow {{x}_{0}}=\dfrac{Mg}{k}\left( 1-\dfrac{AL\sigma }{M} \right)$.
Therefore, the correct option is A.
Note: Note that the direction of the gravitational force on a body is always in the downward direction if the body is on earth as the earth pulls down the body with the gravitational force. The direction of the buoyant force exerted by a liquid is always in the upward direction as it tries to push the body outwards.
Formula used:
${{F}_{g}}=Mg$
${{F}_{B}}={{V}_{s}}g\sigma$
${{F}_{s}}=k{{x}_{0}}$
Complete step by step answer:
At the equilibrium position, the uniform cylinder will be standing straight up.
There will be three forces acting on the cylinder and that are the gravitational force, the spring force and the force of buoyancy exerted by the liquid.

The earth exerts the gravitational force. The gravitational force on a body of mass m is given as ${{F}_{g}}=mg$, where g is the acceleration due to gravity. The gravitational force on the body is always direction downwards.
This means that in the case, the gravitational force on the cylinder is equal to ${{F}_{g}}=Mg$ and is
directed in the downward direction.
When a body is submerged in a liquid, the liquid exerts an upward force on the body called buoyant force. The magnitude of the buoyant force is given as ${{F}_{B}}={{V}_{s}}g\sigma$,
where ${{V}_{s}}$ is the volume of the body submerged in the liquid and $\sigma$ is the density of the liquid.
It is given that half of the cylinder is submerged in the liquid and the volume of a cylinder is equal to $V=AL$, where A is the cross sectional area and L is its length.
This means that ${{F}_{B}}=ALg\sigma$ and is directed upwards.
The cylinder is given to be in equilibrium. Therefore, the cylinder will be at rest and the net force on it will be zero.
Let the extension in the spring be ${{x}_{0}}$. Therefore, the spring force exerted on the cylinder is equal to ${{F}_{s}}=k{{x}_{0}}$, where k is the spring constant of the spring. The spring will be in the upward direction.
Therefore, the net force on the cylinder is ${{F}_{net}}={{F}_{s}}+{{F}_{B}}-{{F}_{g}}$.
$\Rightarrow {{F}_{net}}=k{{x}_{0}}+ALg\sigma -Mg$.
But we know that ${{F}_{net}}=0$.
$\Rightarrow k{{x}_{0}}+ALg\sigma -Mg=0$
$\Rightarrow {{x}_{0}}=\dfrac{Mg-ALg\sigma }{k}$
Take $\dfrac{Mg}{k}$ as a common term.
$\Rightarrow {{x}_{0}}=\dfrac{Mg}{k}\left( 1-\dfrac{AL\sigma }{M} \right)$.
Therefore, the correct option is A.
Note: Note that the direction of the gravitational force on a body is always in the downward direction if the body is on earth as the earth pulls down the body with the gravitational force. The direction of the buoyant force exerted by a liquid is always in the upward direction as it tries to push the body outwards.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




