What is a primary, secondary and tertiary halide?
Answer
537.9k+ views
Hint: Halogens are electronegative in nature and include fluorine, chlorine, bromine and iodine. Alkyl halides are those compounds in which one of the hydrogen atoms of the alkane is replaced with halide. Negative part of the compound is called halide.
Complete answer:
Alkyl halides are those compounds in which one of the hydrogen atoms of the alkane is replaced with halide. The general formula for alkyl halides is \[{{C}_{n}}{{H}_{2n+1}}X\]. Where \[n\] is a natural number. When \[n=1\], then the formula becomes \[C{{H}_{3}}X\] which is called methyl halide. Halides can be fluorides, chlorides, bromide and iodides. In order to form other members of primary halides, one of the hydrogen of methyl halide is replaced with a carbon atom. This is illustrated in the following diagram.
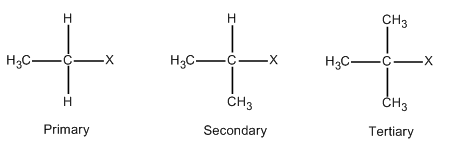
In case of secondary halides, two of the hydrogen of methyl halide is replaced with carbon atoms as described in the above diagram. On the similar lines, three of the hydrogen of methyl halide is replaced with carbon atoms in case of tertiary halides which is also described in the above diagram.
Additional Information: Due to electronegativity difference between halogen and carbon, \[C-X\]bond is polar in nature where \[X\]carries a \[\delta -\] negative charge while alkyl moiety carries a \[\delta +\]charge.
Note:
It is important to note that methyl halide is the simplest primary halide. In order to form other members of primary halides, one of the hydrogen of methyl halide is replaced with a carbon atom. In case of secondary and tertiary halides, two and three of the hydrogen of methyl halides are replaced with carbon atoms, respectively.
Complete answer:
Alkyl halides are those compounds in which one of the hydrogen atoms of the alkane is replaced with halide. The general formula for alkyl halides is \[{{C}_{n}}{{H}_{2n+1}}X\]. Where \[n\] is a natural number. When \[n=1\], then the formula becomes \[C{{H}_{3}}X\] which is called methyl halide. Halides can be fluorides, chlorides, bromide and iodides. In order to form other members of primary halides, one of the hydrogen of methyl halide is replaced with a carbon atom. This is illustrated in the following diagram.

In case of secondary halides, two of the hydrogen of methyl halide is replaced with carbon atoms as described in the above diagram. On the similar lines, three of the hydrogen of methyl halide is replaced with carbon atoms in case of tertiary halides which is also described in the above diagram.
Additional Information: Due to electronegativity difference between halogen and carbon, \[C-X\]bond is polar in nature where \[X\]carries a \[\delta -\] negative charge while alkyl moiety carries a \[\delta +\]charge.
Note:
It is important to note that methyl halide is the simplest primary halide. In order to form other members of primary halides, one of the hydrogen of methyl halide is replaced with a carbon atom. In case of secondary and tertiary halides, two and three of the hydrogen of methyl halides are replaced with carbon atoms, respectively.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

DNA is not present in A Nucleus B Mitochondria C Chloroplast class 11 biology CBSE




