A mixture of ethyl iodide and n-propyl iodide is subjected to wurtz reaction. The hydrocarbon which will be majorly formed is:
A. Butane
B. Propane
C. Pentane
D. Hexane
Answer
608.7k+ views
Hint: Two different or same alkyl halides undergo dimerization in presence of sodium metal is known as Wurtz reaction. This reaction is also called a coupling reaction because the two alkyl halides combine (couples) and form a normal hydrocarbon chain as the product.
Complete step by step answer:
- In the given question the reactants are ethyl iodide and n-propyl iodide.
- The above reactants are subjected to wurtz reaction means reacted in presence of sodium metal.
- The structures of the ethyl iodide and n-propyl iodide are as follows.
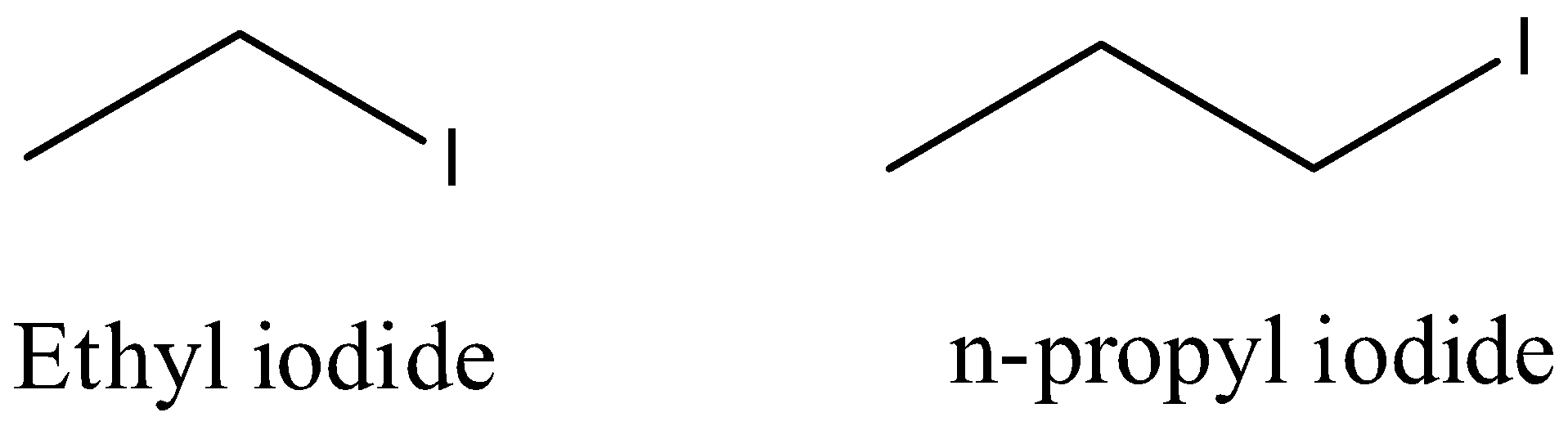
- The reaction of ethyl iodide with n-propyl iodide in presence of sodium metal is as follows.
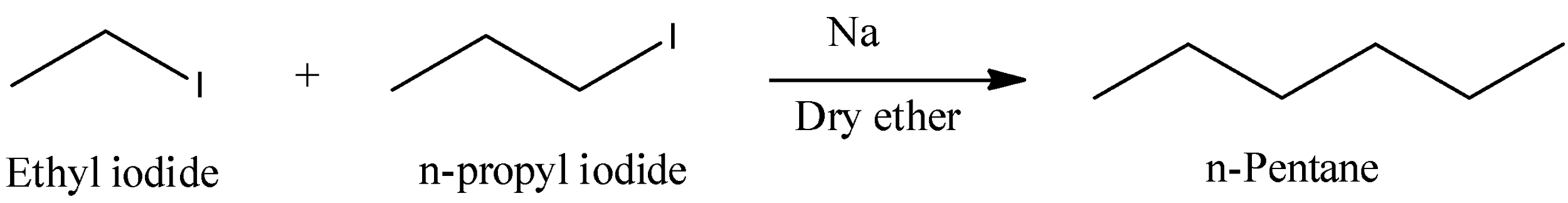
- In the above reaction ethyl iodide couples with n-propyl iodide in presence of sodium metal and forms n-pentane as the product.
- Therefore the product formed when a mixture of ethyl iodide and n-propyl iodide is subjected to wurtz reaction is n-pentane.
So, the correct answer is “Option C”.
Note: In wurtz reaction dry ether acts as a solvent to make the reactants with one another and makes the sodium metal to not react with moisture present in the air. Wurtz reaction has to be carried out in dry medium because sodium metal is highly reactive towards the moisture. Therefore we have to use dehydrated solvents to carry out the wurtz reaction. Generally Wurtz reaction is used to prepare higher alkanes in organic chemistry.
Complete step by step answer:
- In the given question the reactants are ethyl iodide and n-propyl iodide.
- The above reactants are subjected to wurtz reaction means reacted in presence of sodium metal.
- The structures of the ethyl iodide and n-propyl iodide are as follows.
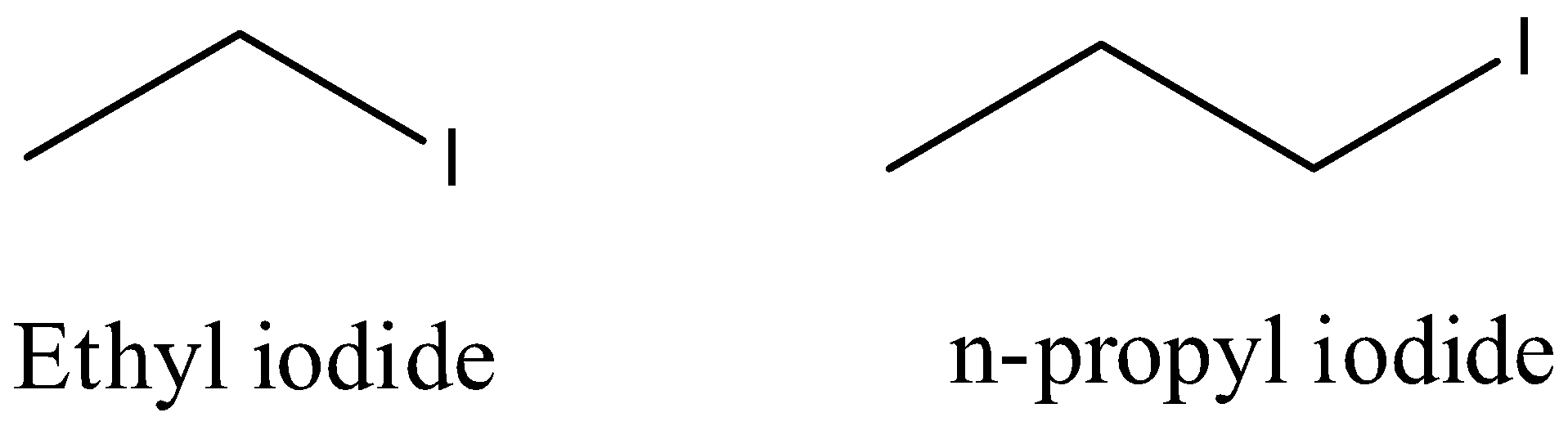
- The reaction of ethyl iodide with n-propyl iodide in presence of sodium metal is as follows.
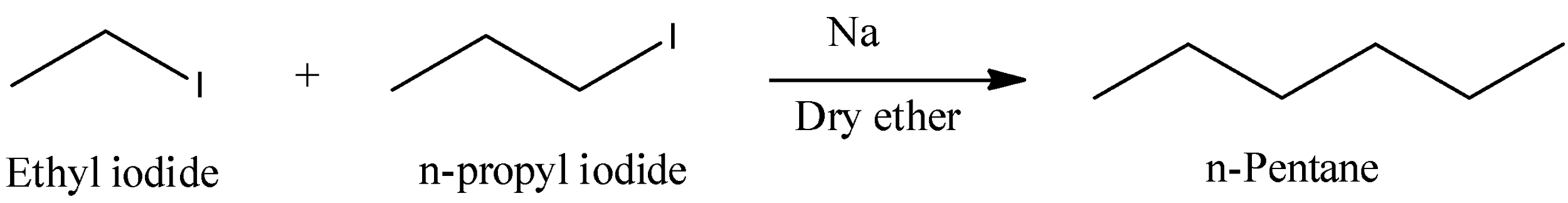
- In the above reaction ethyl iodide couples with n-propyl iodide in presence of sodium metal and forms n-pentane as the product.
- Therefore the product formed when a mixture of ethyl iodide and n-propyl iodide is subjected to wurtz reaction is n-pentane.
So, the correct answer is “Option C”.
Note: In wurtz reaction dry ether acts as a solvent to make the reactants with one another and makes the sodium metal to not react with moisture present in the air. Wurtz reaction has to be carried out in dry medium because sodium metal is highly reactive towards the moisture. Therefore we have to use dehydrated solvents to carry out the wurtz reaction. Generally Wurtz reaction is used to prepare higher alkanes in organic chemistry.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE




