A Fischer projection of (2R,3S)-2,3-butanediol is:
(A)
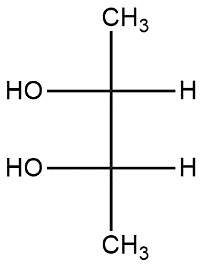
(B)
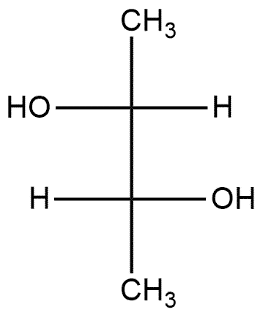
(C)
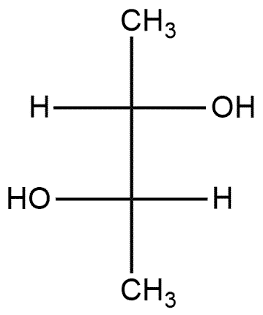
(D)





Answer
606k+ views
Hint: Fischer projection is a two-dimension (2D) representation of a three-dimension (3D) organic molecule with the help of projection. These are mainly used in biochemistry and organic chemistry to represent monosaccharides.
Complete step by step solution:
Stereoisomers are the isomers having similar bonding patterns but differ in the arrangement of atoms or groups in three-dimensional space. There are two different stereoisomers for a molecule that has a centre of chirality in such cases it is expressed in terms of R and S configuration. For any pair of enantiomers with a chirality centre, one will have R configuration and the other will have the S configuration.
R stands for Rectus which is a Latin word with the meaning right, R is given to the isomer when the sequence is left to right.
S stands for Sinister another Latin word with left, S is given to the isomer if the sequence is right to left.
Also if the sequence follows the clockwise pattern it is known to be R configuration while for anticlockwise it follows S configuration. Priority sequence is decided by the sequence rules also called CIP rules.
In the case of 2,3-butanediol, the preference order according to CIP rules is:
\[OH>C{{H}_{3}}>H\]
Now if we see the option B
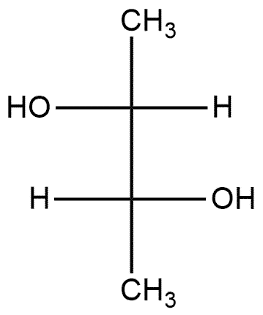
In case of 2nd carbon priority of atoms is started from left to right so it follows R-configuration or on 3rd carbon priority of atoms is started from right to left so it follows S-configuration.
Thus, Fischer projection of (2R,3S)-2,3-butanediol is option (B).
Note: Chiral molecules are those molecules which are not superimposable on their mirror images. Chiral carbon is the carbon which is bonded to different atoms or groups also known by asymmetric carbon.
Complete step by step solution:
Stereoisomers are the isomers having similar bonding patterns but differ in the arrangement of atoms or groups in three-dimensional space. There are two different stereoisomers for a molecule that has a centre of chirality in such cases it is expressed in terms of R and S configuration. For any pair of enantiomers with a chirality centre, one will have R configuration and the other will have the S configuration.
R stands for Rectus which is a Latin word with the meaning right, R is given to the isomer when the sequence is left to right.
S stands for Sinister another Latin word with left, S is given to the isomer if the sequence is right to left.
Also if the sequence follows the clockwise pattern it is known to be R configuration while for anticlockwise it follows S configuration. Priority sequence is decided by the sequence rules also called CIP rules.
In the case of 2,3-butanediol, the preference order according to CIP rules is:
\[OH>C{{H}_{3}}>H\]
Now if we see the option B

In case of 2nd carbon priority of atoms is started from left to right so it follows R-configuration or on 3rd carbon priority of atoms is started from right to left so it follows S-configuration.
Thus, Fischer projection of (2R,3S)-2,3-butanediol is option (B).
Note: Chiral molecules are those molecules which are not superimposable on their mirror images. Chiral carbon is the carbon which is bonded to different atoms or groups also known by asymmetric carbon.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




