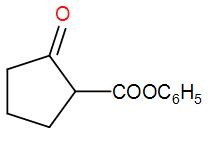
A Dieckmann condensation of diethyl adipate was carried out by heating with sodium ethoxide. One equivalent of benzyl bromide was added, and the resulting mixture was then refluxed in 5% HCl for several hours and extracted with ether. What compound has been prepared by this procedure?
A.
B.
C.
D.




Answer
625.2k+ views
Hint: This is an Intramolecular Nucleophilic Acyl Substitution. In Dieckmann condensation the products are cyclic \[\beta \] – ketoesters and the favoured formation is of the more stable 5 or 6 membered rings. The acyl carbon has a double bond oxygen and an alkyl group, it is a moiety derived by the removal of one or more hydroxyl groups from oxoacids including inorganic acids.
Complete step by step answer:
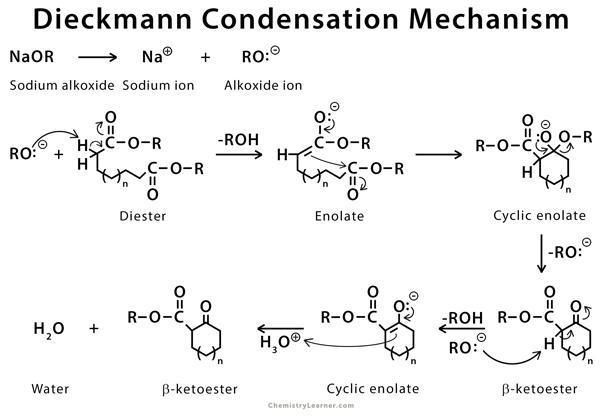
- We can use diester compounds to give an intramolecular Claisen condensation which is also known as Diekmann condensation.
- We use this reaction to form a carbon-carbon bond between two tethered ester groups using an alkoxide base in alcohol to make a cyclic \[\beta \]-keto ester.
- One ester group of the reactant must have an $\alpha $-hydrogen which is abstracted by the base to form an enolate and alcohol.
- The enolate ion attacks the carbonyl carbon of the other ester molecule, to regenerate the base an -OR group is released, and the final product formed is a \[\beta \] – keto ester.
- Now for better understanding let’s see the reaction mechanism:
Sodium ethoxide removes the alpha proton to form enolate ion
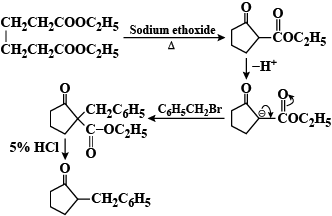
So, the correct answer is “Option B”.
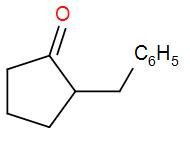
Note: -The most used reagent is alkoxide in Diekmann condensation.
-Remember that enolate ions are good nucleophiles and ester carbonyl carbon atoms are electrophilic.
- Nucleophiles are those reactants which are electron rich and provide a pair of electrons but on contrary electrophiles are electron deficient and accept a pair of electrons to achieve stability.
Complete step by step answer:
- We can use diester compounds to give an intramolecular Claisen condensation which is also known as Diekmann condensation.
- We use this reaction to form a carbon-carbon bond between two tethered ester groups using an alkoxide base in alcohol to make a cyclic \[\beta \]-keto ester.
- One ester group of the reactant must have an $\alpha $-hydrogen which is abstracted by the base to form an enolate and alcohol.
- The enolate ion attacks the carbonyl carbon of the other ester molecule, to regenerate the base an -OR group is released, and the final product formed is a \[\beta \] – keto ester.
- Now for better understanding let’s see the reaction mechanism:
Sodium ethoxide removes the alpha proton to form enolate ion


So, the correct answer is “Option B”.

Note: -The most used reagent is alkoxide in Diekmann condensation.
-Remember that enolate ions are good nucleophiles and ester carbonyl carbon atoms are electrophilic.
- Nucleophiles are those reactants which are electron rich and provide a pair of electrons but on contrary electrophiles are electron deficient and accept a pair of electrons to achieve stability.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




