When $2 - $heptyne was treated with aqueous sulphuric acid containing mercury$(II)$sulphate, two products, each having the molecular formula ${C_7}{H_{14}}O$, were obtained in approximately equal amount. What are these two compounds$?$
$A)$$3 - $heptanone
$B)$heptan$3,4$dione
$C)$$2 - $heptanone
$D)$heptan$3,4$diol
Answer
545.7k+ views
Hint: This is an oxymercuration reaction. Oxymercuration is not limited to an alkene reacting with water. Using an alkyne instead of an alkene yields an enol, which tautomerizes into a ketone. Using alcohol instead of water yields an ether. In both cases, Markovnikov's rule is observed.
Complete answer:
Oxymercuration involves mercury acting as a reagent attacking the alkene double bond to form a mercurinium ion bridge. A water molecule will then attack the most substituted carbon to open the mercurium ion bridge, followed by proton transfer to solvent water molecule.
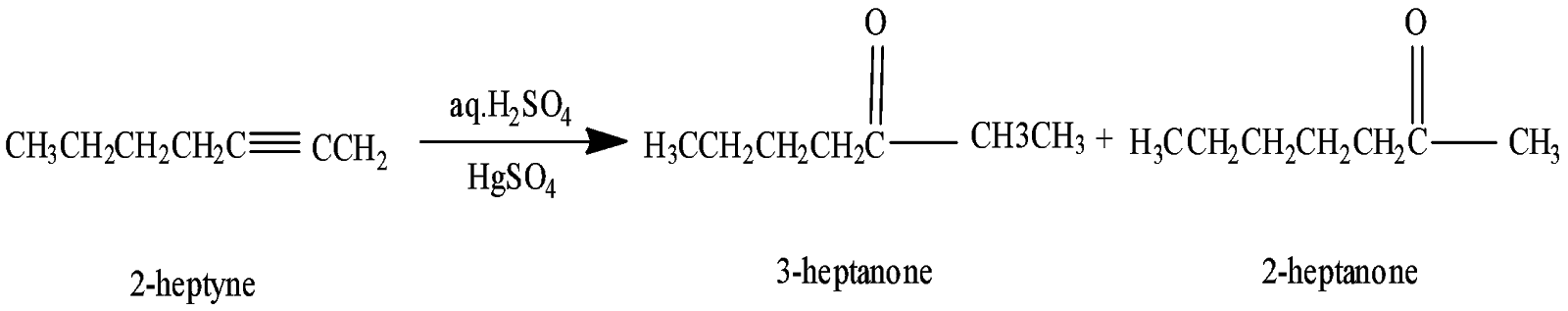
When $2 - $heptyne was treated with aqueous sulphuric acid containing mercury$(II)$ sulphate it gives $3 - heptanone$ and $2 - heptanone$.
Oxymercuration is very regioselective and is a Markovnikov reaction ruling out extreme cases, the water nucleophile will always preferentially attack the more substituted carbon, depositing the resultant hydroxy group there.
$HgS{O_4}$ adds first, in an anti-Markovnikov fashion, forcing a $H$ from ${H_3}{O^ + }$ to add on the terminal carbon and thus water to add to the other side. ${H_2}S{O_4}$ makes the formation of ${H_3}{O^ + }$ more favorable.
So the correct answer is $A)$$3 - $heptanone and $C)$$2 - $heptanone.
Note:
During the oxymercuration reaction the mercury adduct product is almost always treated with sodium borohydride in aqueous base in a reaction called demercuration. In demercuration, the acetyl mercury group is replaced with a hydrogen in a stereochemically insensitive reaction known as reductive elimination.
Complete answer:
Oxymercuration involves mercury acting as a reagent attacking the alkene double bond to form a mercurinium ion bridge. A water molecule will then attack the most substituted carbon to open the mercurium ion bridge, followed by proton transfer to solvent water molecule.

When $2 - $heptyne was treated with aqueous sulphuric acid containing mercury$(II)$ sulphate it gives $3 - heptanone$ and $2 - heptanone$.
Oxymercuration is very regioselective and is a Markovnikov reaction ruling out extreme cases, the water nucleophile will always preferentially attack the more substituted carbon, depositing the resultant hydroxy group there.
$HgS{O_4}$ adds first, in an anti-Markovnikov fashion, forcing a $H$ from ${H_3}{O^ + }$ to add on the terminal carbon and thus water to add to the other side. ${H_2}S{O_4}$ makes the formation of ${H_3}{O^ + }$ more favorable.
So the correct answer is $A)$$3 - $heptanone and $C)$$2 - $heptanone.
Note:
During the oxymercuration reaction the mercury adduct product is almost always treated with sodium borohydride in aqueous base in a reaction called demercuration. In demercuration, the acetyl mercury group is replaced with a hydrogen in a stereochemically insensitive reaction known as reductive elimination.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




