JEE Advanced Updated Chemistry Syllabus 2024: Download the Topic-Wise PDF with Weightage
The revised JEE Advanced 2024 Chemistry Syllabus has been released, and Vedantu brings you the revised JEE Advanced syllabus for Chemistry in the form of downloadable FREE PDFs. It is important to have access to a comprehensive syllabus. It serves as a guide for your preparation, ensuring that all of the important topics are covered.

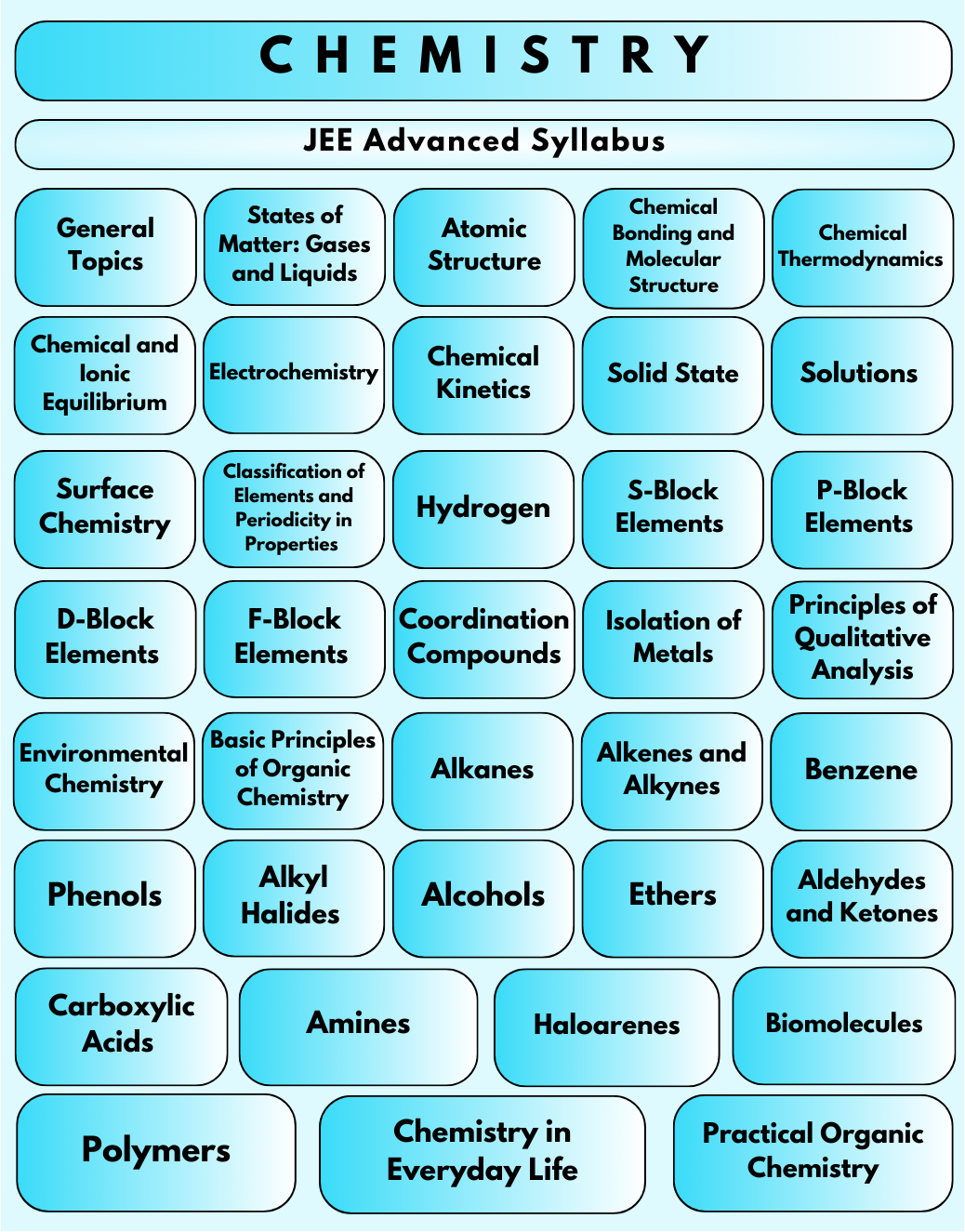
Using Vedantu's syllabus PDF can help you improve your JEE Advanced preparation. The syllabus for JEE Advanced Chemistry is divided into 11 units and covers a wide range of topics such as Inorganic Chemistry, Organic Chemistry, Physical Chemistry and Analytical Chemistry. The PDF also includes information about topic weightage, which can help you prioritise your studies more effectively, ensuring you focus on the most important areas.
USE Coupon Code: CJEE Click Here for Best JEE Crash Course
JEE Advanced Chemistry Syllabus 2024: What's New and What Remains Unchanged?
The JEE Advanced Chemistry syllabus for 2024 has undergone some significant changes, while some topics remain unchanged. The syllabus is based on the latest NCERT textbooks for Class 11 and 12.
Revised Chemistry Syllabus:
The following are some of the minor changes in the revised JEE Advanced Chemistry syllabus:
Subject | New topics Added | Syllabus Reduction |
Chemistry |
|
|
Unlocking More Subjects: JEE Advanced Syllabus 2024 Resources
The JEE Advanced syllabus includes a wide range of Physics, chemistry, and mathematics courses. Because questions frequently need knowledge of numerous areas, it is important to be familiar with the concepts and formulas in all three subjects. By referring to other subjects, students can gain a deeper understanding of the concepts and be more prepared for the JEE Advanced exam. Find links to additional resources that extend beyond the JEE Advanced Chemistry Syllabus 2024.
S.No | Subject-wise JEE Advanced 2024 Syllabus PDF |
1 | |
2 |
JEE Advanced Chemistry Syllabus: 2024 Free PDF Download
Mathematics is extremely important in the JEE Main exam, it serves as a foundation for engineering. The JEE Advanced Chemistry syllabus 2024 covers a wide range of topics and is divided into the following sections:
Inorganic Chemistry
Organic Chemistry
Physical Chemistry
Analytical Chemistry
Each section of the syllabus is further divided into subsections, which covers specific topics. The syllabus is designed to be challenging, but it is also fair. Candidates who have a strong understanding of Chemistry should be able to score well on the JEE Advanced Chemistry exam.
Section A | |
Chapter Name | Topics |
| |
States of Matter: Gases and Liquids |
|
Atomic Structure |
|
| |
Chemical Thermodynamics |
|
| |
| |
| |
Solid State |
|
Solutions |
|
Surface Chemistry |
|
Classification of Elements and Periodicity in Properties |
|
Hydrogen |
|
S-Block Elements |
|
P-Block Elements |
|
D-Block Elements |
|
F-Block Elements |
|
Coordination Compounds |
|
Isolation of Metals |
|
Principles of Qualitative Analysis |
|
Environmental Chemistry |
|
Basic Principles of Organic Chemistry |
|
| |
| |
| |
| |
Alkyl Halides |
|
Alcohols |
|
Ethers |
|
Aldehydes and Ketones |
|
Carboxylic Acids |
|
Amines |
|
Haloarenes |
|
Biomolecules |
|
| |
Chemistry in Everyday Life |
|
Practical Organic Chemistry |
|
Overview of JEE Advanced 2024 Chemistry Topics
The syllabus for JEE Advanced Chemistry typically covers a wide range of topics. To help you navigate through the vast JEE Advanced Chemistry syllabus, we have organized the topics into sections. Here are the key topics that you need to cover:
Inorganic Chemistry
This section covers the fundamental concepts of inorganic chemistry, including chemical bonding, molecular structure, thermodynamics, kinetics, electrochemistry, coordination chemistry, and environmental chemistry.
Chemical Bonding and Molecular Structure: Understanding the different types of chemical bonds, their properties, and how they determine the shape and properties of molecules.
Chemical Thermodynamics: Grasping the principles of thermodynamics, including enthalpy, entropy, and Gibbs free energy, to predict the spontaneity and direction of chemical reactions.
Chemical Kinetics: Comprehending the factors that affect the rate of chemical reactions, including concentration, temperature, and catalysts.
Electrochemistry: Understanding the principles of redox reactions, electrochemical cells, electrode potentials, and electrolysis.
Coordination Chemistry: Learning about the structure, bonding, properties, and applications of coordination compounds.
Environmental Chemistry: Understanding environmental pollution, its sources, and the control measures to mitigate it.
Organic Chemistry
This section focuses on the fundamentals of organic chemistry, encompassing introductory concepts, hydrocarbons, functional groups, organic compounds containing halogens, oxygen, and nitrogen, and biomolecules.
Introductory Organic Chemistry: Understanding the nature, classification, nomenclature, and isomerism of organic compounds.
Hydrocarbons: Grasping the structure, properties, and reactions of aliphatic and aromatic hydrocarbons.
Functional Groups: Understanding the structure, properties, and reactions of various functional groups, including alcohols, ethers, aldehydes, ketones, carboxylic acids, amines, and amides.
Organic Compounds Containing Halogens: Learning about the nomenclature, properties, and reactions of haloalkanes and haloarenes.
Organic Compounds Containing Oxygen: Comprehending the structure, properties, and reactions of organic compounds containing oxygen, such as ethers, aldehydes, ketones, and carboxylic acids.
Organic Compounds Containing Nitrogen: Understanding the structure, properties, and reactions of organic compounds containing nitrogen, particularly amines and amides.
Biomolecules: Learning about the structure, properties, and functions of carbohydrates, lipids, proteins, and nucleic acids.
Physical Chemistry
This section delves into the principles of physical chemistry, covering states of matter, thermodynamics, kinetics, electrochemistry, optics, and modern physics.
States of Matter: Understanding the properties and behavior of matter in the gaseous, liquid, and solid states.
Thermodynamics: Mastering the concepts of thermodynamics, including enthalpy, entropy, Gibbs free energy, and equilibrium.
Kinetics: Grasping the factors that affect the rate of physical processes, including reaction rates, diffusion, and viscosity.
Electrochemistry: Understanding the principles of redox reactions, electrochemical cells, electrode potentials, and electrolysis.
Optics: Comprehending the wave-particle duality of light, reflection, refraction, dispersion, interference, diffraction, and polarization.
Modern Physics: Learning about quantum mechanics, atomic structure, chemical bonding, radioactivity, and nuclear reactions.
Analytical Chemistry
This section focuses on the principles and techniques of analytical chemistry, including basic concepts, gravimetric analysis, volumetric analysis, chromatography, spectroscopy, and instrumental analysis.
Basic Concepts in Analytical Chemistry: Understanding accuracy, precision, errors, calibration, standardization, sampling, and data handling in analytical measurements.
Gravimetric Analysis: Learning about gravimetric techniques, including precipitation gravimetry and volatilization gravimetry.
Volumetric Analysis: Mastering the principles and techniques of volumetric analysis, particularly titrimetric analysis, acid-base titrations, redox titrations, and complexometric titrations.
Chromatography: Understanding the principles and applications of various chromatographic techniques, including paper chromatography, column chromatography, high-performance liquid chromatography (HPLC), and gas chromatography (GC).
Spectroscopy: Comprehending the principles and applications of various spectroscopic techniques, including ultraviolet-visible (UV-Vis) spectroscopy, infrared (IR) spectroscopy, flame photometry, atomic absorption spectroscopy (AAS), and X-ray diffraction (XRD).
Instrumental Analysis: Learning about the principles and applications of advanced instrumental methods of analysis, including UV-Vis spectroscopy, IR spectroscopy, flame photometry, AAS, and XRD.
Weightage of Topics in JEE Advanced 2024 Chemistry
Understanding the importance of each topic in the JEE Advanced 2024 Chemistry syllabus is essential for successful exam preparation. You can better allocate your study time if you know which topics are more important. The following table shows the weightage of each topic:
JEE Advanced Chemistry – Weightage of Chapters | |||
Topics | No of Questions | Marks | Weightage |
Inorganic Chemistry-II | 9 | 33 | 28 |
Organic Chemistry-II | 10 | 32 | 27 |
Physical Chemistry-I | 9 | 24 | 20 |
Physical Chemistry-II | 8 | 23 | 19 |
Aromatic Compounds | 3 | 12 | 10 |
Coordination Compounds | 3 | 11 | 9 |
Qualitative Analysis | 3 | 10 | 8 |
Hydrocarbon (Alkane, Alkene & Alkyne) | 3 | 9 | 8 |
Organic Chemistry-I | 2 | 8 | 7 |
Equivalent Concept | 4 | 8 | 7 |
Thermodynamics and Thermochemistry | 2 | 8 | 7 |
Electrochemistry | 3 | 8 | 7 |
Metallurgy | 1 | 4 | 3 |
p-Block 17-18 Group | 1 | 4 | 3 |
p-block (15-16 Grp) | 1 | 4 | 3 |
Hydrocarbon | 1 | 4 | 3 |
Practical Organic Chemistry | 1 | 4 | 3 |
Biomolecules | 1 | 4 | 3 |
Stereoisomerism | 1 | 3 | 3 |
Aldehyde Ketone | 2 | 4 | 3 |
Atomic Structure & Nuclear Chemistry | 1 | 4 | 3 |
Chemical Equilibrium | 2 | 4 | 3 |
Chemical Kinetics | 1 | 4 | 3 |
Solid State | 1 | 3 | 3 |
Surface Chemistry | 1 | 4 | 3 |
Solution & Colligative Properties | 2 | 4 | 3 |
Total | 38 | 120 | 100 |
By focusing on topics with higher weightage, you can ensure that you cover the most important areas of the syllabus and maximize your chances of scoring well in the exam.
This weightage pattern suggests that Physical Chemistry holds the highest weightage, followed by Organic Chemistry and Inorganic Chemistry. Make sure to take into consideration the entire chapter for a more detailed approach to the topics. However, the topics listed above are the most important ones for the JEE Advanced 2024 Chemistry exam.
Why you should Download JEE Advanced 2024 Chemistry Syllabus PDF?
Chemistry plays a significant role in the JEE Advanced exam. The syllabus is divided into 7 units, each of which is assigned a weightage. The total weightage of the Chemistry syllabus is 30%. The chapters with the highest weightage are Physical Chemistry (40%), Inorganic Chemistry (35%), and Organic Chemistry (25%).
Vedantu's JEE Advanced 2024 Chemistry Syllabus is available in PDF format for free. Simply navigate to the syllabus downloads section, select the Chemistry syllabus, and click to download the PDF file for FREE. This resource will give aspiring candidates a thorough overview of the topics and content they will need to prepare for the upcoming JEE Advanced 2024 examination.
JEE Advanced 2024 Chemistry Syllabus - Best Books for Reference
Preparing for the JEE Advanced 2024 Chemistry exam, which leads to admission to prestigious IITs. Candidates should concentrate their efforts primarily on the topics outlined in the JEE Advanced Chemistry syllabus PDF. It is critical that they refer the appropriate books to aid in their preparation. Here is a list of some of the best JEE Advanced 2024 Chemistry books:
Name of the Book | Author/Publications | Reasons why this Book Stands Out as the Best Choice for JEE Advanced Chemistry |
Physical Chemistry | OP Tandon | Comprehensive coverage of topics, clear explanations, practice problems |
Class 11 and 12 | NCERT books | Official NCERT syllabus coverage, simple and concise explanations |
Modern Approach to Chemical Equations | RC Mukherjee | Focused on problem-solving, step-by-step approach, abundance of practice problems |
Organic Chemistry | OP Tandon | Covers all essential topics, detailed explanations, well-structured |
Organic Chemistry | MS Chauhan | Concept-based approach, variety of solved examples, focus on JEE Advanced |
Organic Chemistry | Peter Sykes | Clear and concise explanations, good for building concepts, numerous practice problems |
Organic Chemistry | Boyd and Morrison | Comprehensive coverage of topics, clear explanations, focus on mechanisms |
Concise Inorganic Chemistry | JD Lee | Concise and to-the-point explanations, covers all essential topics |
Organic Chemistry | Paula Bruice Yurkanis | Concept-based approach, easy-to-understand explanations, relevant examples |
These books cover a wide range of topics and offer detailed explanations, practice problems, and conceptual clarity, making them valuable resources for JEE Advanced 2024 Chemistry prep.
Other Important Links for JEE Advanced Chemistry 2024
Additional resources that go beyond the JEE Advanced 2024 Chemistry Syllabus can be found here. These hyperlinks lead to useful learning materials, practice papers, mock tests, and insights. You can improve your preparation, overcome challenges, and set yourself up for success by using these JEE Advanced 2024 resources. Dive into a world of learning opportunities designed to help you on your way to achieving your exam goals.
Reference Links | Details |
Helps students familiarize themselves with the exam pattern and question types. | |
Allows students to simulate the actual exam environment and assess their time management skills. | |
Provides a comprehensive overview of the syllabus and helps students identify their strengths and weaknesses. | |
Enables students to practice solving past year questions and gain insights into recurring concepts and exam trends. |
Conclusion:
The JEE Advanced Chemistry Syllabus 2024 is a comprehensive guide for students aspiring to crack the prestigious engineering entrance exam. It encompasses a wide range of topics, from Physical Chemistry to Inorganic Chemistry and Organic Chemistry. By staying updated with the syllabus and understanding the weightage of each topic, you can effectively prioritize your study efforts and increase your chances of success in the exam. We encourage you to download the JEE Advanced 2024 Chemistry syllabus PDF and start your exam preparation with confidence.






FAQs on JEE Advanced Chemistry Syllabus 2024 (Released)
1. Is it easy to score full marks in JEE Advanced Chemistry?
Scoring full marks in any subject, including JEE Advanced Chemistry, is extremely challenging. The exam is designed to test the very best students, and the questions are often very difficult and time-consuming. However, it is not impossible to score full marks in JEE Advanced Chemistry. With hard work, dedication, and the right approach, it is achievable.
2. What are the tips to prepare effective notes for JEE Advanced Chemistry?
Here are some tips to prepare effective notes for JEE Advanced Chemistry:
Use your own words: Don't just copy from the textbook or your teacher's notes. Use your own words to explain the concepts.
Be concise: Don't write down everything you hear or read. Focus on the key points and important concepts.
Use diagrams and illustrations: Diagrams and illustrations can help you to understand concepts more clearly.
Organize your notes: Use headings, subheadings, and bullet points to organize your notes.
Review your notes regularly: Review your notes regularly to refresh your memory and identify areas that you need to work on.
3. What are the important chapters to be focussed on in JEE Advanced Chemistry?
While all chapters are important for JEE Advanced Chemistry, some chapters are more heavily weighted than others. Here are some of the most important chapters to focus on:
Physical Chemistry: General Principles and Stoichiometry, States of Matter and Thermodynamics, Chemical Kinetics and Equilibrium
Inorganic Chemistry: Some Basic Concepts of Chemistry, Periodicity and s-Block Elements, p-Block Elements, d-Block Elements
Organic Chemistry: Fundamental Concepts of Organic Chemistry, Hydrocarbons, Organic Compounds Containing Halogen, Alcohol, Ether, Aldehyde, Ketone, Carboxylic Acid, Amines
4. How much time should I dedicate to preparing for JEE Advanced Chemistry?
The amount of time you need to dedicate to preparing for JEE Advanced Chemistry will vary depending on your starting point and your goals. However, it is generally recommended to start studying early and dedicate at least 3-4 hours per day to chemistry.
5. What are the challenges of preparing for JEE Advanced Chemistry?
The JEE Advanced Chemistry exam is a challenging exam that requires a significant amount of hard work and dedication. Some of the challenges of preparing for the exam include:
The vast amount of content to cover
The difficulty of the questions
The competition from other top-performing students



